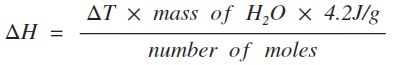|
Section 4: Physical chemistry
|
a) Acids, alkalis and salts
4.1: Describe the use of the indicators litmus, phenolphthalein, and methyl orange to distinguish between acidic and alkaline solutions
Litmus
When red litmus is inserted in an acidic solution, it stays red
When red litmus is inserted in an alkaline solution, it turns blue
When blue litmus is inserted in an acidic solution, it turns red
When blue litmus is inserted in an alkaline solutions, it stays blue
Phenolphthalein
When phenolphthalein is inserted in an acidic solution, it becomes transparent
When phenolphthalein is inserted in an alkaline solution, it stays pink
Methyl Orange
When methyl orange is inserted in an acidic solution, it turns red
When methyl orange is inserted in an alkaline solution, it turns yellow
Litmus
When red litmus is inserted in an acidic solution, it stays red
When red litmus is inserted in an alkaline solution, it turns blue
When blue litmus is inserted in an acidic solution, it turns red
When blue litmus is inserted in an alkaline solutions, it stays blue
Phenolphthalein
When phenolphthalein is inserted in an acidic solution, it becomes transparent
When phenolphthalein is inserted in an alkaline solution, it stays pink
Methyl Orange
When methyl orange is inserted in an acidic solution, it turns red
When methyl orange is inserted in an alkaline solution, it turns yellow
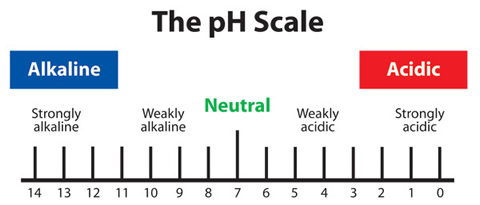
4.2: Understand how the pH scale, from 0-14, can be used to classify solutions as strongly acidic, weakly acidic, neutral, weakly alkaline, or strongly alkaline
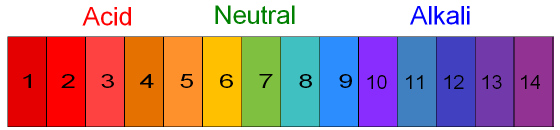
4.3: Describe the use of universal indicator to measure the approximate pH value of a solution
Like litmus paper, universal indicator can also be used to measure the pH value of an acid or alkaline solution. But rather than turning red or blue, universal indicator will turn into one of a variety of colors that determines how acidic or alkaline the solution is:
Like litmus paper, universal indicator can also be used to measure the pH value of an acid or alkaline solution. But rather than turning red or blue, universal indicator will turn into one of a variety of colors that determines how acidic or alkaline the solution is:
4.4: Define acids as sources of hydrogen ions, H+, and alkalis as sources of hydroxide ions, OH-
When acids are dissolved in water, they produce hydrogen ions, for example, when hydrochloric acid is dissolved in water, the products are hydrogen and chloride ions
When alkalis are dissolved in water, they produce hydroxide ions, for example, when sodium hydroxide is dissolved in water, the products are sodium and hydroxide ions
When acids are dissolved in water, they produce hydrogen ions, for example, when hydrochloric acid is dissolved in water, the products are hydrogen and chloride ions
When alkalis are dissolved in water, they produce hydroxide ions, for example, when sodium hydroxide is dissolved in water, the products are sodium and hydroxide ions
4.5: Predict the products of reactions between dilute hydrochloric, nitric and sulfuric acids; and metals, metal oxides and metal carbonates (excluding the reactions between nitric acid and metals)
|
Metal hydroxides (mostly soluble)
acid + metal hydroxide → salt + water
Metal oxides (mostly soluble) acid + metal oxide → salt + water
|
Metal carbonates (mostly insoluble)
acid + metal carbonate → salt + water + carbon dioxide
Metals acid + metal → salt + hydrogen
|
|
4.6: Understand the general rules for predicting the solubility of salts in water:
|
Understand the general rules for predicting state symbols in chemical equations:
|
4.7: Describe experiments to prepare soluble salts from acids
Excess Metal/Base
Excess Metal/Base
- Add an excess of solid metal to a beaker of acid and stir
- Add more metal until the reaction is complete (i.e. all the acid is gone)
- Filter off the excess metal
- Heat the solution; the solution will be saturated when crystals form on a glass rod dipped into the solution
- Stop heating and allow solution to cool once saturated
- Filter again to remove water
- Dry the crystals with filter paper or by placing on a warm gauze mat
|
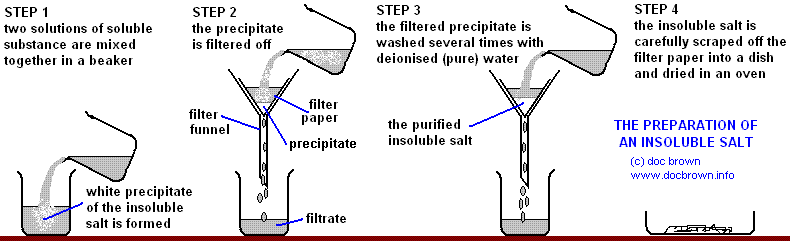
4.8: Describe experiments to prepare insoluble salts using precipitation reactions
Precipitation
|
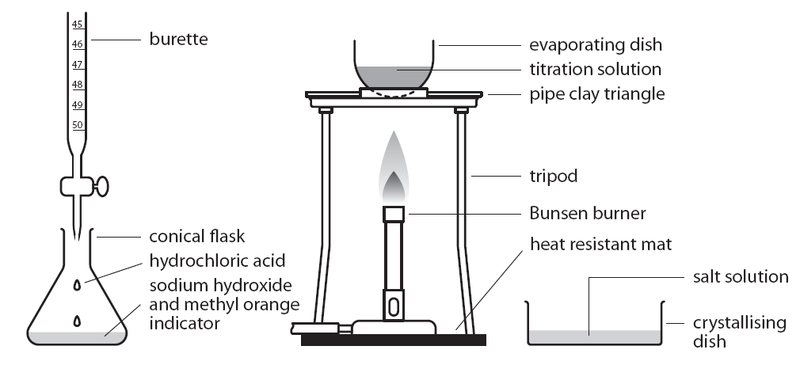
4.9: Describe experiments to carry out acid-alkali titrations
Titration
Titration
- Add the acid to the alkali and indicator using a burette until the solution is neutral (or until temperature stops rising)
- Measure the volume of acid that has been added; throw away the solution as it contains the indicator
- Add the same volume of acid to the same amount of alkali without indicator
- Heat the solution; the solution will be saturated when crystals form on a glass rod dipped into the solution
- Stop heating and allow solution to cool once saturated
- Filter again to remove water
- Dry the crystals with filter paper or by placing on a warm gauze mat
b) Energetics
4.10: Recall that chemical reactions in which heat energy is given out are described as exothermic and those in which heat energy is taken in are endothermic
An exothermic reaction is one which releases heat energy. There is an observable rise in temperature (e.g. when a test tube gets warmer the reaction is exothermic). As energy is released to the surroundings, the temperature of the surroundings increases. Exothermic reactions need a small amount of energy to get started but once one has started it goes on and does not need to be given any energy anymore as it provides its own. Most reactions are exothermic and some important ones include:
An endothermic reaction is one which absorbs energy from the surroundings. The energy is usually in the form of heat or light. As they system removes energy from the surroundings, the temperature of the surroundings decreases. An endothermic needs a continuous input of energy - when you take away the heat or light source the reaction stops. Important endothermic reactions include:
An exothermic reaction is one which releases heat energy. There is an observable rise in temperature (e.g. when a test tube gets warmer the reaction is exothermic). As energy is released to the surroundings, the temperature of the surroundings increases. Exothermic reactions need a small amount of energy to get started but once one has started it goes on and does not need to be given any energy anymore as it provides its own. Most reactions are exothermic and some important ones include:
- burning of fossil fuels - fuel + oxygen → carbon dioxide + water
- respiration - glucose + oxygen → carbon dioxide + water
- neutralization reactions
- electrochemical reactions
An endothermic reaction is one which absorbs energy from the surroundings. The energy is usually in the form of heat or light. As they system removes energy from the surroundings, the temperature of the surroundings decreases. An endothermic needs a continuous input of energy - when you take away the heat or light source the reaction stops. Important endothermic reactions include:
- photosynthesis - carbon dioxide + water → glucose + oxygen
- decomposition reactions like electrolysis
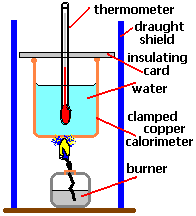
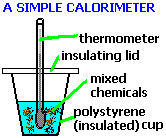
4.11: Describe simple calorimetry experiments for reactions, such as combustion, displacement, dissolving and neutralization in which heat energy changes can be calculated from measured temperature changes
4.12: Calculate molar enthalpy change from heat energy change
The amount of energy released or absorbed by a reaction can be measured experimentally using this equation:
The amount of energy released or absorbed by a reaction can be measured experimentally using this equation:
In this equation:
- ΔH is the molar enthalpy change, measured in J/mol or kJ/mol
- ΔT is the temperature increase or decrease caused by the reaction, measured in °C
- mass of H2O is the mass of water, measured in grams
- 4.2J/g is the specific heat capacity of water
4.13: Understand the use of ΔH to represent molar enthalpy change for exothermic and endothermic reactions
ΔH is the amount of heat energy or enthalpy absorbed or released for one mole of a substance; it is called the molar enthalpy change. It is measured in J/mol or kJ/mol.
ΔH is the amount of heat energy or enthalpy absorbed or released for one mole of a substance; it is called the molar enthalpy change. It is measured in J/mol or kJ/mol.
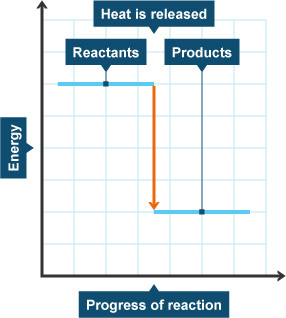
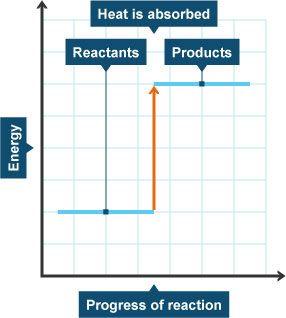
4.14: Represent exothermic and endothermic reactions on a simple energy level diagram
Energy changes taking place during reactions can be shown in energy level diagrams. The horizontal lines show us how much energy the reactants and products have:
Energy changes taking place during reactions can be shown in energy level diagrams. The horizontal lines show us how much energy the reactants and products have:
|
In an exothermic reaction:
|
In an endothermic reaction:
|
4.15: Recall that the breaking of bonds is endothermic and that the making of bonds is exothermic
During any chemical reaction the particles are rearranged. For this to happen, bonds within the reactants need to be broken. When bonds are broken new bonds can be made. Energy must be supplied to break old bonds, and energy is released when new bonds form. If in a reaction more energy is released for making new bonds than what is needed to break the old ones, the reaction is exothermic and ΔH will have a negative value. In an endothermic reaction, more energy is needed to break the bonds than the amount released when new ones are made. Therefore, ΔH will be positive.
During any chemical reaction the particles are rearranged. For this to happen, bonds within the reactants need to be broken. When bonds are broken new bonds can be made. Energy must be supplied to break old bonds, and energy is released when new bonds form. If in a reaction more energy is released for making new bonds than what is needed to break the old ones, the reaction is exothermic and ΔH will have a negative value. In an endothermic reaction, more energy is needed to break the bonds than the amount released when new ones are made. Therefore, ΔH will be positive.
4.16: Use average bond energies to calculate the enthalpy change during a simple chemical reaction
If we know how much energy is needed to break bonds and how much energy is released when new bonds are made we can calculate the overall enthalpy change and find out if the reaction is endothermic or exothermic. Bond energies can be used to carry out such calculations. The bond energy of a covalent bond is the amount of energy needed to break one mole of that bond; it is also the amount of energy released when that bond is made. For example:
The ΔH of the reaction = the sum of bond energies of reactants - the sum of bond energies of products
ΔH of reaction = 2648kJ - 3348kJ = -700kJ
If we know how much energy is needed to break bonds and how much energy is released when new bonds are made we can calculate the overall enthalpy change and find out if the reaction is endothermic or exothermic. Bond energies can be used to carry out such calculations. The bond energy of a covalent bond is the amount of energy needed to break one mole of that bond; it is also the amount of energy released when that bond is made. For example:
- CH4 + 2O2 → CO2 + 2H2O
The ΔH of the reaction = the sum of bond energies of reactants - the sum of bond energies of products
- Bonds broken = (4 x (C-H) + 2 x (O=O)) = (4 x 413) + (2 x 498) = 2648kJ
- Bonds made = (2 x (C=O) + 4 x (H-O)) = (2 x 746) + (4 x 464) = 3348kJ
ΔH of reaction = 2648kJ - 3348kJ = -700kJ
c) Rates of reaction
4.17: Describe experiments to investigate the effects of changes in surface area of a solid, concentration of solutions, temperature and the use of a catalyst on the rate of reaction
Surface Area
Surface Area
- React an acid and a metal - time how long it takes for the metal to disappear
- Crush the metal in order to powder it - time how long it takes for the metal to disappear
- React an acid and a metal - time how long it takes for the metal to disappear
- Dilute the acid before reacting it with the metal - time how long it takes for the metal to disappear
- React an acid and a metal - time how long it takes for the metal to disappear
- Heat the acid before reacting it with the metal - time how long it takes for the metal to disappear
- React an acid and a metal - time how long it takes for the metal to disappear
- Redo the reaction but add a catalyst - time how long it takes for the metal to disappear
4.18: Describe the effects of changes in surface area of a solid, concentration of solutions, pressure of gases, temperature and the use of a catalyst on the rate of a reaction
Surface area: As the surface area of the reactant gets larger, the rate of reaction increases
Concentration: As the concentration of the acid gets higher, the rate of reaction increases
Pressure of gases: As the pressure of the gases gets higher, the rate of reaction increases
Temperature: As the temperature of the reactant gets higher, the rate of reaction increases
Catalyst: If there is a catalyst added to a reaction, the rate of the reaction increases
Surface area: As the surface area of the reactant gets larger, the rate of reaction increases
Concentration: As the concentration of the acid gets higher, the rate of reaction increases
Pressure of gases: As the pressure of the gases gets higher, the rate of reaction increases
Temperature: As the temperature of the reactant gets higher, the rate of reaction increases
Catalyst: If there is a catalyst added to a reaction, the rate of the reaction increases
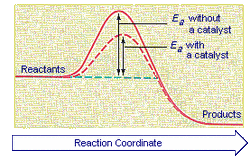
4.19: Understand the term activation energy and represent it on a reaction profile
Activation energy is the amount of energy required for a reaction to start happening
Activation energy is the amount of energy required for a reaction to start happening
Ea = Activation Energy
4.20: Explain the effects of changes in surface area of a solid, concentration of solutions, pressure of gases and temperature on the rate of a reaction in terms of particle collision theory
Surface Area
When the surface area of a reactant increases, there are more particles available and therefore there is potential for more successful collisions to occur between the reactant and the solution
Concentration
If the particles in the solution are more concentrated, they will have more energy and therefore the collisions will faster
Pressure of Gases
If the reactants are two gases, the more pressurized they are, the more successful collisions will occur (similar to concentration)
Temperature
If the particles in the solution are at a higher temperature, they have more kinetic energy and therefore the collisions will be faster
Surface Area
When the surface area of a reactant increases, there are more particles available and therefore there is potential for more successful collisions to occur between the reactant and the solution
Concentration
If the particles in the solution are more concentrated, they will have more energy and therefore the collisions will faster
Pressure of Gases
If the reactants are two gases, the more pressurized they are, the more successful collisions will occur (similar to concentration)
Temperature
If the particles in the solution are at a higher temperature, they have more kinetic energy and therefore the collisions will be faster
4.21: Explain that a catalyst speeds up a reaction by providing an alternative pathway with lower activation energy
Catalysts work by bringing the reactive parts of reactant particles closer to each other, and thus provide a pathway with lower activation energy needed than would be required in a regular reaction. More of the reactants will posses this lower activation energy, and therefore the reactants will happen faster
Catalysts work by bringing the reactive parts of reactant particles closer to each other, and thus provide a pathway with lower activation energy needed than would be required in a regular reaction. More of the reactants will posses this lower activation energy, and therefore the reactants will happen faster
d) Equilibria
4.22: Recall that some reactions are reversible and are indicated by the symbol ⇌ in equations
In some chemical reactions, the products can be changed back into reactants
Chemical reactions in which products can be turned back into reactants are called reversible reactions and they are indicated by the symbol ⇌.
In some chemical reactions, the products can be changed back into reactants
- The reaction form reactant into products is called the forward reaction
- The reaction which changes the products back into reactants is called the reverse reaction
Chemical reactions in which products can be turned back into reactants are called reversible reactions and they are indicated by the symbol ⇌.
4.23: Describe reversible reactions such as the dehydration of copper(II) sulfate and the effect of heat on ammonium chloride
Dehydrating by thermal decomposition and hydrating copper sulfate is a reversible reaction:
When we heat blue hydrated copper sulfate, it decomposes into white anhydrous copper sulfate and water. We can now get the hydrated copper sulfate back by either allowing the products to cool as the anhydrous copper sulfate absorbs water from the air to change back into blue hydrated copper sulfate or adding water to the white anhydrous copper sulfate. This releases heat as it is exothermic. The original change has been exactly reversed. Even the heat that was originally put in has been given out again. Heating the blue hydrated copper sulfate again will again bring back the anhydrous white copper sulfate.
Another reversible reaction is the thermal decomposition of ammonium chloride:
Heating ammonium chloride favors the forward reaction and the white powder decomposes into two colorless gases, NH3 and HCl. Allowing the mixture of the two gases to cool causes them to form ammonium chloride again which is what happens in the cooler parts of the test tube. The reversible reaction is a neutralization reaction as it involves an acid and an alkali.
Dehydrating by thermal decomposition and hydrating copper sulfate is a reversible reaction:
- CuSO4•5H2O ⇌ CuSO4 + 5H2O
When we heat blue hydrated copper sulfate, it decomposes into white anhydrous copper sulfate and water. We can now get the hydrated copper sulfate back by either allowing the products to cool as the anhydrous copper sulfate absorbs water from the air to change back into blue hydrated copper sulfate or adding water to the white anhydrous copper sulfate. This releases heat as it is exothermic. The original change has been exactly reversed. Even the heat that was originally put in has been given out again. Heating the blue hydrated copper sulfate again will again bring back the anhydrous white copper sulfate.
Another reversible reaction is the thermal decomposition of ammonium chloride:
- NH4Cl ⇌ HCl + NH3
Heating ammonium chloride favors the forward reaction and the white powder decomposes into two colorless gases, NH3 and HCl. Allowing the mixture of the two gases to cool causes them to form ammonium chloride again which is what happens in the cooler parts of the test tube. The reversible reaction is a neutralization reaction as it involves an acid and an alkali.
|
4.24: Understand the concept of dynamic equilibrium
Reversible reactions under closed conditions can reach dynamic equilibrium. Closed means that no substances are added to the reaction mixture and no substances escape form it. However, heat can escape or be absorbed. In a reversible reaction, both the forward and reverse reaction go on at the same time. This means that in such a system we always have reactant particles as well as product particles. The forward reaction is always the first reaction to happen but as soon as some product is formed the reverse reaction will start. At some point during the reaction, a steady state is reached, when both the forward and reverse reactions will be going on at the same speed or rate. This means that the reactants are now being changed into products as fast as the products are changed back into reactants. As a result the amounts of reactant and product now remains the same. It is equilibrium in the sense that the total amounts of the various things present are now constant. When this is the case, dynamic equilibrium is reached. At dynamic equilibrium, the amount of reactants and products stays the same. There appears to be no change. However, the amount of reactants and products are not necessarily equal to each other. |
4.25: Predict the effects of changing the pressure and temperature on the equilibrium position in reversible reactions
Chemists are interested in how much reactant and how much product is present at equilibrium. The relative amounts of each is indicated by referring to the position of equilibrium. The position of equilibrium tells us how the amounts of reactant and product compare with each other.
For industrial processes, it is important to maximize the amount of the desired chemical. If your desired chemical is a product of a reversible reaction, you don't want it all changing back into a reactant as soon as it is made. The position of equilibrium can be changed by making the forward reaction go faster or changing the conditions of the reaction. Making changes to the pressure (in reactions involving gases) or temperature alters the position of equilibrium. Le Chatelier's principle says that the reaction will counteract any changes you make to it, including temperature:
This is important for industrial processes such as the Haber process. The forward reaction, which makes ammonia for fertilizers, is exothermic. So the yield of ammonia is better at lower temperatures. However, if the reaction is at too low a temperature, then it happens at a much slower rate. This would make the process uneconomical. A compromise temperature is chosen: one that is high enough to get a reasonable rate of reaction, but not so high that the yield of ammonia is low.
Pressure is caused by molecules hitting the walls of their container - the more molecules, the greater the pressure. If gases are present in the equilibrium mixture, then a change in the pressure may affect the position of equilibrium:
There is a limit to the pressure that can be used industrially, because very high pressures require very strong and expensive equipment. This means a compromise pressure is chosen - high enough to get a good yield but not so high that it would add much to the cost of the process.
Chemists are interested in how much reactant and how much product is present at equilibrium. The relative amounts of each is indicated by referring to the position of equilibrium. The position of equilibrium tells us how the amounts of reactant and product compare with each other.
- If there is more product than reactant, we say the equilibrium is towards the products, towards the right or that the forward reaction is favored. In most reactions this is what we want.
- If there is more reactant than product, we say the equilibrium is towards the reactants, towards the left or that the reverse reaction is favored. This is not usually what we want.
For industrial processes, it is important to maximize the amount of the desired chemical. If your desired chemical is a product of a reversible reaction, you don't want it all changing back into a reactant as soon as it is made. The position of equilibrium can be changed by making the forward reaction go faster or changing the conditions of the reaction. Making changes to the pressure (in reactions involving gases) or temperature alters the position of equilibrium. Le Chatelier's principle says that the reaction will counteract any changes you make to it, including temperature:
- If the forward reaction is exothermic and the temperature is increased, the yield of products is decreased. If the temperature is decreased, the yield of products is increased.
- If the forward reaction is endothermic and the temperature is increased, the yield of products is increased. If the temperature is decreased, the yield of products is decreased.
- Increasing the temperature always favors the endothermic reaction.
- Decreasing the temperature always favors the exothermic reaction.
This is important for industrial processes such as the Haber process. The forward reaction, which makes ammonia for fertilizers, is exothermic. So the yield of ammonia is better at lower temperatures. However, if the reaction is at too low a temperature, then it happens at a much slower rate. This would make the process uneconomical. A compromise temperature is chosen: one that is high enough to get a reasonable rate of reaction, but not so high that the yield of ammonia is low.
Pressure is caused by molecules hitting the walls of their container - the more molecules, the greater the pressure. If gases are present in the equilibrium mixture, then a change in the pressure may affect the position of equilibrium:
- If the pressure is increased, the position of equilibrium moves in the direction of the fewest molecules
- If the pressure is decreased, the position of equilibrium moves in the direction of the most molecules
- Changes in pressure will not affect the position of equilibrium if the number of moles of gases on both sides of the equation are equal
- Adding a catalyst does not affect the position of equilibrium because both forward and reverse reactions are sped up
There is a limit to the pressure that can be used industrially, because very high pressures require very strong and expensive equipment. This means a compromise pressure is chosen - high enough to get a good yield but not so high that it would add much to the cost of the process.