|
Section 5: Chemistry in industry
|
a) Extraction and uses of metals
5.1: Explain how the methods of extraction of the metals in this section are related to their positions in the reactivity series
The ease of obtaining a pure metal from its ore depends on the reactivity of the metal. A metal ore is a mixture of rock which contains a compound with the metal in it. The less reactive metals are easily removed from their ores as it only takes heating to do this. More reactive metals are difficult to obtain as their oxides or salts are difficult to decompose so a lot of energy is needed, which is why they can only be reduced using electrolysis. The unreactive metals do not need extraction by electrolysis as they are found pure in nature, although copper is electrolyzed further as it needs to be very pure to be used as a conductor of electricity.
The ease of obtaining a pure metal from its ore depends on the reactivity of the metal. A metal ore is a mixture of rock which contains a compound with the metal in it. The less reactive metals are easily removed from their ores as it only takes heating to do this. More reactive metals are difficult to obtain as their oxides or salts are difficult to decompose so a lot of energy is needed, which is why they can only be reduced using electrolysis. The unreactive metals do not need extraction by electrolysis as they are found pure in nature, although copper is electrolyzed further as it needs to be very pure to be used as a conductor of electricity.
|
5.2: Describe and explain the extraction of aluminum from purified aluminum oxide by electrolysis
Aluminum ore is also known as bauxite, a rock which mainly contains aluminum oxide but also has some impurities like iron oxide and silica. There are several conditions that need to be met for the extraction form aluminum from bauxite:
Aluminum ore is also known as bauxite, a rock which mainly contains aluminum oxide but also has some impurities like iron oxide and silica. There are several conditions that need to be met for the extraction form aluminum from bauxite:
- The electrolytic cell needs to have carbon electrodes and the carbon cathode needs line the outside of the cell rather than be in the solution itself like the anode.
- The aluminum oxide needs to be heated until it is molten in order to carry out electrolysis; no external heat is required during electrolysis as the large current releases enough heat energy to keep the aluminum oxide molten.
- A substance called cryolite is added to the mixture to lower the mixture's overall melting point, so less energy is required to electrolyze the aluminum oxide. The molten cryolite also acts as a solvent.
- Very large amounts of current are required, which is why most aluminum smelters are near power stations.
- After some time, the positive carbon electrodes need to be replaced, as they react with oxygen, producing carbon dioxide and wearing away at the electrodes. These two conditions make the process very expensive.
5.3: Write ionic half-equations for the reactions at the electrodes in aluminum extraction
|
Reaction at the anode
Carbon dioxide is formed when the oxygen reacts with the carbon electrode. This reaction is made easier by the large amount of heat energy. The carbon electrodes get worn away and need to be replaced periodically. Half-equation at the anode 2O2- → O2 + 4e- |
Reaction at the cathode
Positively charged molten aluminum is attracted to the negatively charged cathode. This reaction depends on the large amount of electrical current and the molten cryolite that lowers the overall operating temperature. Half-equation at the cathode Al3+ + 3e- → Al |
|
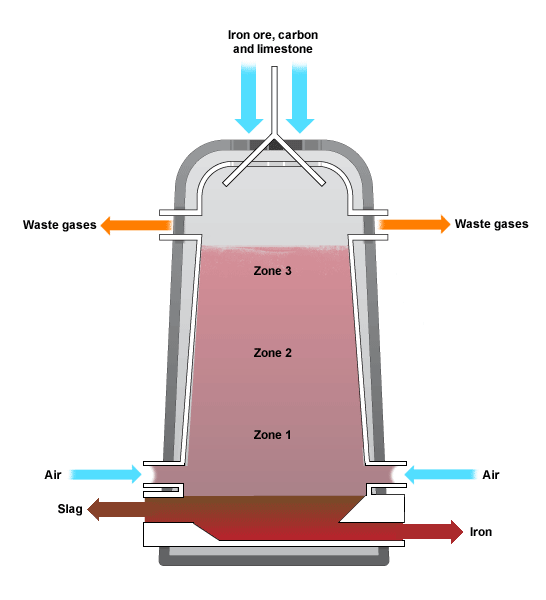
5.4: Describe and explain the main reactions involved in the extraction of iron from iron ore
Iron extraction is carried out in a blast furnace using the following raw materials:
There are five main reaction which go on in a blast furnace:
|
5.5: Explain the uses of aluminum and iron, in terms of their properties
|
Uses of aluminum
|
Uses of iron alloys
|
b) Crude oil
5.6: Understand that crude oil is a mixture of hydrocarbons
Crude oil is a mixture of hydrocarbons.
Crude oil is a mixture of hydrocarbons.
5.7: Describe and explain how the industrial process of fractional distillation separates crude oil into fractions
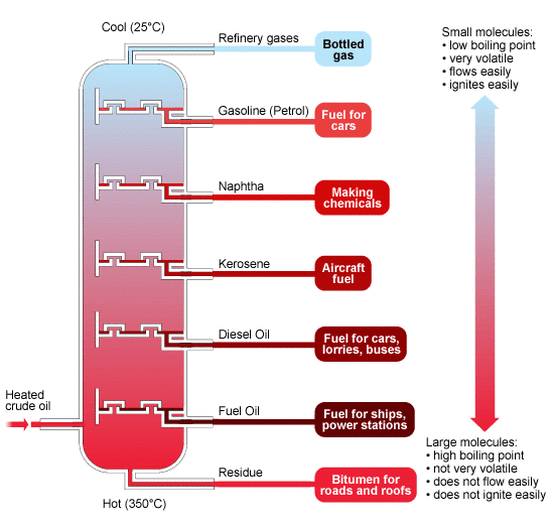
Crude oil, as such, has no direct use. It has to be refined before it is any use. The first step in the refining of crude oil is fractional distillation.
Fractional distillation is carried out in a fractionating column. The column is hot at the bottom and gradually becomes cooler at the top.
The crude oil is split into various fractions as described in the diagram. A fraction is a mixture of hydrocarbons with very similar boiling points.
Crude oil is heated to convert it into a vapour. The vapour is then fed into the bottom of the fractionating column.
The hydrocarbons with very high boiling points (fuel, oil and bitumen) immediately turn into liquids and are tapped off at the bottom of the column.
Fractional distillation is carried out in a fractionating column. The column is hot at the bottom and gradually becomes cooler at the top.
The crude oil is split into various fractions as described in the diagram. A fraction is a mixture of hydrocarbons with very similar boiling points.
Crude oil is heated to convert it into a vapour. The vapour is then fed into the bottom of the fractionating column.
The hydrocarbons with very high boiling points (fuel, oil and bitumen) immediately turn into liquids and are tapped off at the bottom of the column.
5.8: Recall the names and uses of the main fractions obtained from crude oil: refinery gases, gasoline, kerosene, diesel, fuel oil and bitumen
|
• Refinery gases are used for bottled gas, which is butane or propane stored in portable tanks
• Gasoline is used for fuel in cars • Kerosene is used for aircraft fuel • Diesel oil is used as fuel in cars, lorries, and buses • Fuel oil is used as fuel in ships and power stations • Residue is used for bitumen in roads and roofs |
Robin Gets Killed Dead From Batman
|
5.9: Describe the trend in boiling point and viscosity of the main fractions
As boiling point goes up, viscosity goes up.
As boiling point goes up, viscosity goes up.
5.10: Understand that incomplete combustion of fuels may produce carbon monoxide and explain that carbon monoxide is poisonous because it reduces the capacity of the blood to carry oxygen
Incomplete combustion (combustion where not enough oxygen is available for complete combustion) may result in the poisonous gas carbon monoxide being produced. When inhaled, carbon monoxide will combine with oxygen carrying haemoglobin and it will cause the haemoglobin to no longer be available to carry oxygen. This will prevent the oxygenated blood from reaching vital organs like the heart and brain.
Incomplete combustion (combustion where not enough oxygen is available for complete combustion) may result in the poisonous gas carbon monoxide being produced. When inhaled, carbon monoxide will combine with oxygen carrying haemoglobin and it will cause the haemoglobin to no longer be available to carry oxygen. This will prevent the oxygenated blood from reaching vital organs like the heart and brain.
5.11: Understand that, in car engines, the temperature reached is high enough to allow nitrogen and oxygen from air to react, forming nitrogen oxide
In an active car engine, the temperatures can get so high that the nitrogen and oxygen inside the engine can react to form nitrogen oxides.
In an active car engine, the temperatures can get so high that the nitrogen and oxygen inside the engine can react to form nitrogen oxides.
5.12: Understand that nitrogen oxides and sulfur dioxide are pollutant gases which contribute to acid rain, and describe the problems caused by acid rain
Nitrogen oxides and sulfur dioxide are both pollutant gases which contribute to acid rain. Acid rain can get into water sources and poison the animals living in them, it can destroy trees, plants and forests, and can also damage structures and cause them to break.
Nitrogen oxides and sulfur dioxide are both pollutant gases which contribute to acid rain. Acid rain can get into water sources and poison the animals living in them, it can destroy trees, plants and forests, and can also damage structures and cause them to break.
5.13: Understand that fractional distillation of crude oil produces more long-chain hydrocarbons than can be used directly and fewer short-chain hydrocarbons than required and explain why this makes cracking necessary
Crude oil is a mixture of hydrocarbons - it can be separated by fractional distillation. Larger hydrocarbons are broken down into smaller hydrocarbons by cracking. Cracking is when a long-chain hydrocarbon is put into a catalytic cracker with an absence of air, high temperatures (600-700°C), and a catalyst of aluminum oxide to speed up the process. The result is a mixture of alkanes and alkenes. Smaller hydrocarbon molecules like these are less viscous and more flammable. They are more useful (i.e. petrol is more in demand than diesel) and cracking also produces a by-product that is used to make plastic.
Crude oil is a mixture of hydrocarbons - it can be separated by fractional distillation. Larger hydrocarbons are broken down into smaller hydrocarbons by cracking. Cracking is when a long-chain hydrocarbon is put into a catalytic cracker with an absence of air, high temperatures (600-700°C), and a catalyst of aluminum oxide to speed up the process. The result is a mixture of alkanes and alkenes. Smaller hydrocarbon molecules like these are less viscous and more flammable. They are more useful (i.e. petrol is more in demand than diesel) and cracking also produces a by-product that is used to make plastic.
5.14: Describe how long-chain alkanes are converted to alkenes and shorter-chain alkanes by catalytic cracking, using silica or alumina as the catalyst and a temperature in the range of 600-700°C
Long-chain alkenes are broken down into smaller hydrocarbons in a catalytic cracker. Alumina or silica is used as a catalyst in the conditions of an absence of air as well as a temperature range from 600-700°C.
Long-chain alkenes are broken down into smaller hydrocarbons in a catalytic cracker. Alumina or silica is used as a catalyst in the conditions of an absence of air as well as a temperature range from 600-700°C.
c) Synthetic polymers
5.16: Draw the repeat unit of addition polymers, including poly(ethene), poly(propene) and poly (chloroethene)
5.17: Deduce the structure of monomer from the repeat unit of an addition polymer
An addition polymer forms bonds only across double bonds.
An addition polymer forms bonds only across double bonds.
5.18: Describe some uses for polymers, including poly(ethene), poly(propene), and poly(chloroethene)
1. Poly(ethene)
Poly(ethene)is resistant to chemical attack and can be used to store food, drinks and chemicals.
High density poly(ethene) is used in milk jugs, detergent bottles, margarine tubs, and garden furniture.
Medium density poly(ethene) is used in gas pipes, rubbish bins, and storage tanks for fuel.
Low density poly(ethene) is used for plastic bags and cling film.
2. Poly(propene)
Poly(propene) is tougher and harder wearing than poly(ethene). It is used for making food packaging, ropes, and carpets.
3. Poly(chloroethene)
Poly(chloroethene) is tougher than poly(ethene), very hard wearing, and more stable to heat. It can be used for plastic sheets, artificial leather, drainpipes, gutters, insulation for electrical wires and casings for electrical plugs.
1. Poly(ethene)
Poly(ethene)is resistant to chemical attack and can be used to store food, drinks and chemicals.
High density poly(ethene) is used in milk jugs, detergent bottles, margarine tubs, and garden furniture.
Medium density poly(ethene) is used in gas pipes, rubbish bins, and storage tanks for fuel.
Low density poly(ethene) is used for plastic bags and cling film.
2. Poly(propene)
Poly(propene) is tougher and harder wearing than poly(ethene). It is used for making food packaging, ropes, and carpets.
3. Poly(chloroethene)
Poly(chloroethene) is tougher than poly(ethene), very hard wearing, and more stable to heat. It can be used for plastic sheets, artificial leather, drainpipes, gutters, insulation for electrical wires and casings for electrical plugs.
5.19: Explain that addition polymers are hard to dispose of as their inertness means that they do not easily biodegrade
Addition polymers are inert (chemically inactive) which means that they do not easily biodegrade because they are unreactive or their reactions are extremely slow.
Addition polymers are inert (chemically inactive) which means that they do not easily biodegrade because they are unreactive or their reactions are extremely slow.
5.20: Understand that some polymers, such as nylon, form by a different process called condensation polymerization
Some polymers like nylon form by a process called condensation polymerization. It involves a series of condensation reactions between two monomers. Each monomer normally contains two functional groups.
Some polymers like nylon form by a process called condensation polymerization. It involves a series of condensation reactions between two monomers. Each monomer normally contains two functional groups.
5.21: Understand that condensation polymerization produces a small molecule, such as water, as well as the polymer
A condensation reaction is a chemical reaction in which two molecules combine to form a larger molecule (the polymer) with the elimination of a smaller molecule such as water or ammonia.
A condensation reaction is a chemical reaction in which two molecules combine to form a larger molecule (the polymer) with the elimination of a smaller molecule such as water or ammonia.
d) The industrial manufacture of chemicals
5.21: Recall that nitrogen from air, and hydrogen from natural gas or the cracking of hydrocarbons, are used in the manufacture of ammonia
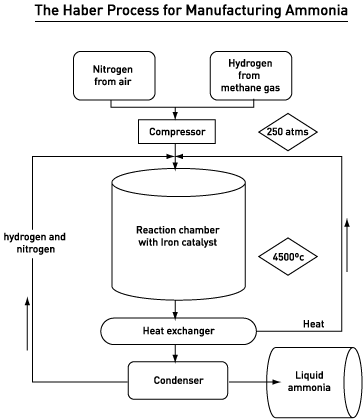
The starting reactants in the Haber process are hydrogen obtained from natural gas or cracking, and nitrogen from air.
The starting reactants in the Haber process are hydrogen obtained from natural gas or cracking, and nitrogen from air.
5.22: Describe the manufacture of ammonia by the Haber process, including the essential conditions
There are essential conditions when manufacturing ammonia to ensure high yield:
There are essential conditions when manufacturing ammonia to ensure high yield:
- a temperature of 450°C
- a pressure of 200 atmospheres
- an iron catalyst
5.23: Understand how the cooling of the reaction mixture liquifies the ammonia produced and allows the unused hydrogen and nitrogen to be recirculated
The manufacture of ammonia is a reversible reaction so the reaction chamber always contains a mixture of hydrogen, nitrogen and ammonia. The ammonia is separated as the reaction mixture is pumped to the condenser where it is allowed to cool. Ammonia has a higher boiling point than nitrogen and hydrogen so it condenses out and is collected as a liquid; the gaseous nitrogen and hydrogen are recycled and pumped back into the reaction chamber.
The manufacture of ammonia is a reversible reaction so the reaction chamber always contains a mixture of hydrogen, nitrogen and ammonia. The ammonia is separated as the reaction mixture is pumped to the condenser where it is allowed to cool. Ammonia has a higher boiling point than nitrogen and hydrogen so it condenses out and is collected as a liquid; the gaseous nitrogen and hydrogen are recycled and pumped back into the reaction chamber.
5.24: Recall the use of ammonia in the manufacture of nitric acid and fertilizers
Ammonia is used to make fertilizers such as ammonium nitrate and other ammonium compounds. It is also used in the manufacture of nitric acid.
Ammonia is used to make fertilizers such as ammonium nitrate and other ammonium compounds. It is also used in the manufacture of nitric acid.
5.25: Recall the raw materials used in the manufacture of sulfuric acid
The raw materials used in the manufacture of sulfuric acid are sulfur and air.
The raw materials used in the manufacture of sulfuric acid are sulfur and air.
5.26: Describe the manufacture of sulfuric acid by the contact process, including the essential conditions
There are essential conditions when manufacturing sulfuric acid to ensure high yield:
- The burning of sulfur: S + O2 → SO2
- The burning of sulfur dioxide in oxygen to make sulfur trioxide: 2SO2 + O2 ⇋ 2SO3
- The dissolving of the sulfur trioxide in concentrated sulfuric acid to form oleum: SO3 + H2SO4 → H2S2O7
- The reaction of oleum with water to form sulfuric acid: H2S2O7 + H2O → H2SO4
There are essential conditions when manufacturing sulfuric acid to ensure high yield:
- a temperature of 450°C - the oxidation of sulfur dioxide into sulfur trioxide is an exothermic process. To favor the forward exothermic reaction the temperature should be low, but like in the Haber process, the temperature is a compromise between percentage yield of sulfur trioxide and the rate of the reaction
- a pressure of 1-2 atmospheres - the product side as fewer moles than the reactant side so increasing the pressure favors the forward reaction. The pressure does not need to be so high as in the Haber process as the yield is already high at normal pressure.
- a vanadium oxide catalyst - speeds up the reaction as it lowers the activation energy
5.27: Recall the use of sulfuric acid in the manufacture of detergents, fertilizers and paints
Sulfuric acid is used to make fertilizers, detergents and soaps. It can also be used in the manufacture of paint.
Sulfuric acid is used to make fertilizers, detergents and soaps. It can also be used in the manufacture of paint.
5.28: Describe the manufacture of sodium hydroxide and chlorine by the electrolysis of concentrated sodium chloride solution (brine) in a diaphragm cell
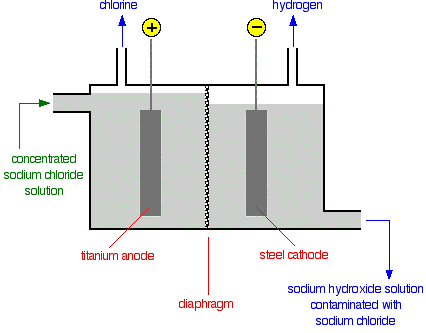
At the start of the reaction, sodium and hydrogen ions will be present at the steel cathode, and chlorine and hydroxide ions at the titanium anode. The products of the reaction must be kept apart - chlorine and hydrogen would explode. A porous membrane is kept between the electrodes; the membrane prevents the chlorine and sodium hydroxide from reacting to form bleach as the solution can only flow from the anode to the cathode. There is more solution on the left (anode) side - this ensures flow of liquid from left to right to prevent sodium hydroxide going near chlorine.
At the start of the reaction, sodium and hydrogen ions will be present at the steel cathode, and chlorine and hydroxide ions at the titanium anode. The products of the reaction must be kept apart - chlorine and hydrogen would explode. A porous membrane is kept between the electrodes; the membrane prevents the chlorine and sodium hydroxide from reacting to form bleach as the solution can only flow from the anode to the cathode. There is more solution on the left (anode) side - this ensures flow of liquid from left to right to prevent sodium hydroxide going near chlorine.
5.29: Write ionic half-equations for the reactions at the electrodes in the diaphragm cell
Ionic half-equation for the reaction at the cathode:
Ionic half-equation for the reaction at the anode:
Ionic half-equation for the reaction at the cathode:
- 2H+ + 2e- → H2
Ionic half-equation for the reaction at the anode:
- 2Cl- → Cl2 + 2e-
5.30: Recall important uses of sodium hydroxide, including the manufacture of bleach, paper and soap; and of chlorine, including sterilizing water supplies and in the manufacture of bleach and hydrochloric acid
Sodium hydroxide is used in the manufacture of bleach, paper and soap. Chlorine is used to sterilize water supplies, manufacture bleach and hydrochloric acid.
Sodium hydroxide is used in the manufacture of bleach, paper and soap. Chlorine is used to sterilize water supplies, manufacture bleach and hydrochloric acid.