|
Section 7: Radioactivity and particles
|
a) Units
7.1: Use the following units: becquerel (Bq), centimeter (cm), hour (h), minute (min), second (s)
Becquerel: Used to measure how many unstable nuclei are disintegrating per second
Centimeter: Used to measure distance
Hour: Used to measure time
Minute: Used to measure time
Second: Used to measure time
Becquerel: Used to measure how many unstable nuclei are disintegrating per second
Centimeter: Used to measure distance
Hour: Used to measure time
Minute: Used to measure time
Second: Used to measure time
b) Radioactivity
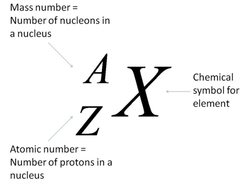
7.2: Describe the structure of an atom in terms of protons, neutrons and electrons and use atomic notation to describe particular nuclei
The nucleus of an atom contains positively charged protons and neutral neutrons. The nucleus is surrounded by smaller, negatively charged particles called electrons. Each element has its own atomic number, which is the number of protons in the nucleus, and the mass number, which is the number of nucleons (protons and neutrons) in the nucleus:
The nucleus of an atom contains positively charged protons and neutral neutrons. The nucleus is surrounded by smaller, negatively charged particles called electrons. Each element has its own atomic number, which is the number of protons in the nucleus, and the mass number, which is the number of nucleons (protons and neutrons) in the nucleus:
7.3: Understand the terms atomic (proton) number, mass (nucleon) number and isotope
- The atomic (proton) number of an element is the number of protons in atom of the element
- The mass (nucleon) number of an element is the number of nucleons (protons and neutrons) in an atom of the element
- Isotopes of an element are atoms with a different number of neutrons in the nucleus
7.4: Understand that alpha and beta particles and gamma rays are ionizing radiations emitted from unstable nuclei in a random process
Isotopes, which are radioactive forms of some elements, will decay randomly over time. They can emit different types of radiation. Gamma radiation has not effect on the atomic mass or charge of the decaying atom but can appear alongside other types of radiation. The different types of ionizing radiation are alpha (α), beta (β) particles, and gamma (Ɣ) rays.
Isotopes, which are radioactive forms of some elements, will decay randomly over time. They can emit different types of radiation. Gamma radiation has not effect on the atomic mass or charge of the decaying atom but can appear alongside other types of radiation. The different types of ionizing radiation are alpha (α), beta (β) particles, and gamma (Ɣ) rays.
7.5: Describe the nature of alpha and beta particles and gamma rays and recall that they may be distinguished in terms of penetrating power
Ionizing radiation is emitted from unstable nuclei. Alpha particles are helium nuclei ejected from unstable nuclei. They are heavily ionizing and have only a short range, traveling only ~10cm in air. They can easily be stopped by thin card.
Beta particles are fast moving electrons ejected from unstable nuclei. They are less ionizing and travel long distances in air. They are stopped by 1-2mm of aluminum.
Gamma rays are photons of high energy EM waves. They are extremely penetrating and interact with atoms which may then emit ionizing radiation. They can only be stopped by tens of cm of lead.
Ionizing radiation is emitted from unstable nuclei. Alpha particles are helium nuclei ejected from unstable nuclei. They are heavily ionizing and have only a short range, traveling only ~10cm in air. They can easily be stopped by thin card.
Beta particles are fast moving electrons ejected from unstable nuclei. They are less ionizing and travel long distances in air. They are stopped by 1-2mm of aluminum.
Gamma rays are photons of high energy EM waves. They are extremely penetrating and interact with atoms which may then emit ionizing radiation. They can only be stopped by tens of cm of lead.
7.6: Describe the effects on the atomic and mass numbers of a nucleus of the emission of each of the three main types of radiation
Both alpha and beta emissions cause a change in the atomic number of the original decaying element - in both alpha and beta decay processes the original element turns into another element.
An isotope of the radioactive element americium is used in some types of smoke detector. It decays by emitting an alpha particle. An atom of americium-241 decays to an atom of neptunium-237 and emits an alpha particle (a helium atom).
A radioactive isotope of sodium decays to magnesium by emitting a beta particle (an electron). An atom of sodium-26 decays to an atom of magnesium and emits a beta particle.
Both alpha and beta emissions cause a change in the atomic number of the original decaying element - in both alpha and beta decay processes the original element turns into another element.
An isotope of the radioactive element americium is used in some types of smoke detector. It decays by emitting an alpha particle. An atom of americium-241 decays to an atom of neptunium-237 and emits an alpha particle (a helium atom).
A radioactive isotope of sodium decays to magnesium by emitting a beta particle (an electron). An atom of sodium-26 decays to an atom of magnesium and emits a beta particle.
7.7: Understand how to complete balanced nuclear equations
Equations which show radioactive decay must balance. The sum of the atomic masses before and after the decay process must be the same. the sum of the atomic numbers must be the same before and after the decay.
Equations which show radioactive decay must balance. The sum of the atomic masses before and after the decay process must be the same. the sum of the atomic numbers must be the same before and after the decay.
7.8: Understand that ionizing radiations can be detected using a photographic film or a Geiger-Muller detector
Nuclear radiation can ionize atoms that it interacts with. Nuclear radiation can be detected in two main ways. Photographic film becomes fogged when exposed to ionizing radiation and is used in badges worn by workers at risk from continuous exposure. The badges are checked regularly to ensure that safety limits have not been exceeded. The Geiger-Müller tube can also be used to detect ionizing radiation. When radiation enters the GM tube, it ionizes the gases within allowing a pulse of current to pass between the electrodes. This is then fed to either a counter or a rate meter. Often the current pulses are made to produce audible click sounds.
Nuclear radiation can ionize atoms that it interacts with. Nuclear radiation can be detected in two main ways. Photographic film becomes fogged when exposed to ionizing radiation and is used in badges worn by workers at risk from continuous exposure. The badges are checked regularly to ensure that safety limits have not been exceeded. The Geiger-Müller tube can also be used to detect ionizing radiation. When radiation enters the GM tube, it ionizes the gases within allowing a pulse of current to pass between the electrodes. This is then fed to either a counter or a rate meter. Often the current pulses are made to produce audible click sounds.
7.9: Understand the sources of background radiation
There are several sources of natural radiation. The slow decay of isotopes of uranium in the Earth’s rocks produces the gases radon and thoron. Radon is highly radioactive and is a particular problem in some parts of the UK.
When stars explode, the very violent reactions produce cosmic rays which shower the Earth. Radioactive materials are used in diagnosis and treatment of illnesses. These contribute to background radiation. Testing of nuclear weapons contributes a small amount of background radiation. Leaks from nuclear power stations are also responsible for a small percentage of the background count.
There are several sources of natural radiation. The slow decay of isotopes of uranium in the Earth’s rocks produces the gases radon and thoron. Radon is highly radioactive and is a particular problem in some parts of the UK.
When stars explode, the very violent reactions produce cosmic rays which shower the Earth. Radioactive materials are used in diagnosis and treatment of illnesses. These contribute to background radiation. Testing of nuclear weapons contributes a small amount of background radiation. Leaks from nuclear power stations are also responsible for a small percentage of the background count.
7.10: Understand that the activity of a radioactive source decreases over a period of time and is measured in becquerels
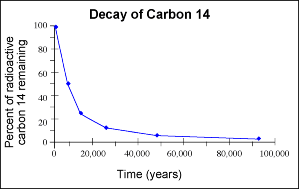
Radioactive decay happens randomly. It is impossible to predict when a particular atom will decay. However, previous measurements taken can be used to make predictions about what percentage of unstable atoms in a sample will decay in a given time. The rate of decay is measured in becquerels - one Bq is one decay per second.
The number of unstable nuclei gets smaller as decay proceeds so the rate of decay also becomes smaller. The proportion of the material that decays in a given time does not change.
Radioactive decay happens randomly. It is impossible to predict when a particular atom will decay. However, previous measurements taken can be used to make predictions about what percentage of unstable atoms in a sample will decay in a given time. The rate of decay is measured in becquerels - one Bq is one decay per second.
The number of unstable nuclei gets smaller as decay proceeds so the rate of decay also becomes smaller. The proportion of the material that decays in a given time does not change.
|
7.11: Understand the term 'half-life' and understand that it is different for different radioactive isotopes
The radioactive half-life of an isotope is the time taken for half the original number of unstable nuclei to decay. The half-life does not change. However, different isotopes have different half-lives from each other. For example, uranium-238 has a half-life of 4.5 billion years, and the half-life of sodium-24 is about 15 hours. |
7.12: Use the concept of half-life to carry out simple calculations on activity
7.13: Describe the uses of radioactivity in medical and non-medical tracers, in radiotherapy, and in the radioactive dating of archaeological specimens and rocks
Radioactive tracers are used to monitor the function of parts of the body. Chemicals containing emitters of gamma radiations are swallowed or injected and these follow the tracer through the digestive system or veins and arteries. Some compounds are chosen because they have a higher concentration in particular organs, allowing the organs to be examined closely.
Radioactive compounds can also directly be used to treat illness. For example, iodine-131, a beta emitter, is swallowed and treats a condition of the thyroid gland. The iodine is concentrated in the thyroid and the radiation kills cells. Focused beams of gamma radiation also kill cancer cells.
Microorganisms on surgical equipment and food can be killed with ionizing radiation. The items to be sterilized can be sealed (in airtight packaging) and can be sterilized through the packaging if more penetrating radiation is used.
The flow of liquids and gases through industrial processes can be mapped using radioactive tracers and detectors. Gamma cameras can also be used.
The proportion of the radioactive isotope carbon-14 in samples of dead organic material can be used to date how long the material has been dead. Carbon-14 is chemically identical to carbon-12, which is not radioactive. It is absorbed by all living things during respiration. The proportion of carbon-14 to carbon-12 reduces predictably after death. Some inorganic materials can be dated by measuring the proportion of a radioactive isotope relative to the material formed at the end of its decay chain. Radioactive materials go through a series of decays, turning into lighter elements. The end product is a stable isotope of the element.
Radioactive tracers are used to monitor the function of parts of the body. Chemicals containing emitters of gamma radiations are swallowed or injected and these follow the tracer through the digestive system or veins and arteries. Some compounds are chosen because they have a higher concentration in particular organs, allowing the organs to be examined closely.
Radioactive compounds can also directly be used to treat illness. For example, iodine-131, a beta emitter, is swallowed and treats a condition of the thyroid gland. The iodine is concentrated in the thyroid and the radiation kills cells. Focused beams of gamma radiation also kill cancer cells.
Microorganisms on surgical equipment and food can be killed with ionizing radiation. The items to be sterilized can be sealed (in airtight packaging) and can be sterilized through the packaging if more penetrating radiation is used.
The flow of liquids and gases through industrial processes can be mapped using radioactive tracers and detectors. Gamma cameras can also be used.
The proportion of the radioactive isotope carbon-14 in samples of dead organic material can be used to date how long the material has been dead. Carbon-14 is chemically identical to carbon-12, which is not radioactive. It is absorbed by all living things during respiration. The proportion of carbon-14 to carbon-12 reduces predictably after death. Some inorganic materials can be dated by measuring the proportion of a radioactive isotope relative to the material formed at the end of its decay chain. Radioactive materials go through a series of decays, turning into lighter elements. The end product is a stable isotope of the element.
7.14: Describe the dangers of ionizing radiations
Ionizing radiations may cause mutations in living organisms. Mutations are random changes in the genetic structure of an organism which cause cells to behave differently, sometimes leading to uncontrollable cell division which leads to cancer. Prolonged exposure to ionizing radiation can also cause damage to cells and tissue.
There are precautions that can be taken to protect oneself from the potential dangers of ionizing radiation. Workers in the nuclear industry wear badges to indicate their level of exposure. Some of these badges are strips of photographic film that become increasingly foggy as exposure increases. Another type of badge uses a property called thermoluminescence, which means that the badge will give out light when it is exposed to high levels of radiation. Samples of radioactive material should be stored in lead containers and in bunkers for prolonged storage, as some materials can be active for thousands of years.
Ionizing radiations may cause mutations in living organisms. Mutations are random changes in the genetic structure of an organism which cause cells to behave differently, sometimes leading to uncontrollable cell division which leads to cancer. Prolonged exposure to ionizing radiation can also cause damage to cells and tissue.
There are precautions that can be taken to protect oneself from the potential dangers of ionizing radiation. Workers in the nuclear industry wear badges to indicate their level of exposure. Some of these badges are strips of photographic film that become increasingly foggy as exposure increases. Another type of badge uses a property called thermoluminescence, which means that the badge will give out light when it is exposed to high levels of radiation. Samples of radioactive material should be stored in lead containers and in bunkers for prolonged storage, as some materials can be active for thousands of years.
c) Particles
7.15: Describe the results of Geiger and Marsden's experiments with gold foil and alpha particles
Geiger and Marsden carried out a series of experiments which involved firing alpha particles at gold foil. These experiments proved the existence of the nucleus. The experiment they carried out had the following procedure: they surrounded gold foil by a zinc sulphide screen, which gave out scintillations when hit by an alpha particle. Most alpha particles passed through the gold foil as if it wasn’t there. Some were knocked off course. Very few bounced off the gold. This proved that most of each atom must be empty space, and the alpha particles which were deflected bounced off something with a large mass and tiny volume: the nucleus.
Geiger and Marsden carried out a series of experiments which involved firing alpha particles at gold foil. These experiments proved the existence of the nucleus. The experiment they carried out had the following procedure: they surrounded gold foil by a zinc sulphide screen, which gave out scintillations when hit by an alpha particle. Most alpha particles passed through the gold foil as if it wasn’t there. Some were knocked off course. Very few bounced off the gold. This proved that most of each atom must be empty space, and the alpha particles which were deflected bounced off something with a large mass and tiny volume: the nucleus.
7.16: Describe Rutherford's nuclear model of the atom and how it accounts for the results of Geiger and Marsden's experiment and understand the factors which affect the deflection of alpha particles by a nucleus
The amount of deflection in the experiment depended on a number of factors:
Analysis of the results of the experiment allowed the prediction of the size of the nucleus.
The amount of deflection in the experiment depended on a number of factors:
- the speed of the alpha particle - the alpha particle is deflected less if it is traveling faster
- the nuclear charge - if the nucleus is strongly positive, then the alpha particle will be more strongly repelled away from it
- how close the alpha particle gets to the positively charged nucleus
Analysis of the results of the experiment allowed the prediction of the size of the nucleus.
7.17: Understand that a nucleus of U-235 can be split (the process of fission) by collision with a neutron, and that this process releases energy in the form of kinetic energy of the fission products
The isotope uranium-235 is fissile - it can be split into lighter elements quite easily. If an atom of U-235 is struck by a neutron it breaks apart, releasing energy in the form of the kinetic energy of the decay fragments.
The isotope uranium-235 is fissile - it can be split into lighter elements quite easily. If an atom of U-235 is struck by a neutron it breaks apart, releasing energy in the form of the kinetic energy of the decay fragments.
7.18: Understand that the fission of U-235 produces two daughter nuclei and a small number of neutrons
The parent nucleus, U-235, produces two daughter nuclei plus a number of neutrons.
The parent nucleus, U-235, produces two daughter nuclei plus a number of neutrons.
7.19: Understand that a chain reaction can be set up if the neutrons produced by one fission strike other U-235 nuclei
If neutrons produced during the fission of U-235 are absorbed by further fissile nuclei, a chain reaction results with rapidly increasing numbers of atoms splitting apart and releasing energy - such a reaction takes place inside a nuclear bomb.
If neutrons produced during the fission of U-235 are absorbed by further fissile nuclei, a chain reaction results with rapidly increasing numbers of atoms splitting apart and releasing energy - such a reaction takes place inside a nuclear bomb.
7.20: Understand the role played by the control rods and moderator when the fission process is used as an energy source to generate electricity
If the chain reaction in a fissile nuclear fuel like uranium-235 is controlled, so that the energy is released much more slowly, the heat produced can be used to generate steam to turn turbines and drive generators. The graphite moderator is used to absorb some of the energy of fast neutrons so that they are readily absorbed by nuclei of U-235 sustaining the chain reaction. The boron control rods absorb neutrons to take them out of action completely. The control rods can be raised out of the reactor core allowing the chain reaction to speed up, or lowered completely to shut down the chain reaction.
If the chain reaction in a fissile nuclear fuel like uranium-235 is controlled, so that the energy is released much more slowly, the heat produced can be used to generate steam to turn turbines and drive generators. The graphite moderator is used to absorb some of the energy of fast neutrons so that they are readily absorbed by nuclei of U-235 sustaining the chain reaction. The boron control rods absorb neutrons to take them out of action completely. The control rods can be raised out of the reactor core allowing the chain reaction to speed up, or lowered completely to shut down the chain reaction.