|
Section 1: Principles of chemistry
|
a) States of matter
1.1: Understand the arrangement, movement and energy of particles in each of the three states of matter: solid, liquid and gas
Solids
• Particles are close together and touching one another
• Particles are arranged in a regular, repeating pattern
• Particles vibrate in their fixed positions but do not move apart
• The forces between the particles are stronger than in liquids
Example: Ice
Liquids
• Particles are close together and touching one another
• Particles are arranged in an irregular pattern
• Particles move around and slide past one another
• The forces between the particles are not as strong as in solids
Example: Water
Gases
• Particles are far apart
• Particles are arranged in an irregular pattern
• Particles move freely and collide with one another
• The forces between the particles are non-existent
Example: Water Vapor
Solids
• Particles are close together and touching one another
• Particles are arranged in a regular, repeating pattern
• Particles vibrate in their fixed positions but do not move apart
• The forces between the particles are stronger than in liquids
Example: Ice
Liquids
• Particles are close together and touching one another
• Particles are arranged in an irregular pattern
• Particles move around and slide past one another
• The forces between the particles are not as strong as in solids
Example: Water
Gases
• Particles are far apart
• Particles are arranged in an irregular pattern
• Particles move freely and collide with one another
• The forces between the particles are non-existent
Example: Water Vapor
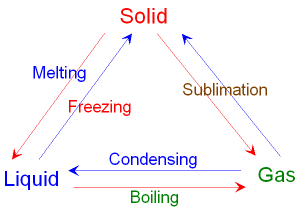
1.2: Understand how the interconversions of solids, liquids, and gases are achieved and recall the names used for these interconversions
|
1.3: Explain the changes in arrangement, movement and energy of particles during these conversions
Melting (solid to liquid)
The particles gain kinetic energy and vibrate faster and faster. This allows the particles to overcome the forces of attraction that hold them together in the solid. The regular pattern is broken down and the particles can now slide past one another.
Freezing (liquid to solid)
The particles lose kinetic energy and this allows the forces of attraction between the particles to hold them together. The particles arrange themselves into a regular pattern and are no longer able to slide past one another.
Boiling (liquid to gas)
The particles gain kinetic energy and move further apart. Eventually the forces of attraction between the particles are completely broken and they are able to escape from the liquid.
Condensation (gas to liquid)
The particles lose kinetic energy and this allows the forces of attraction to tiring the particles closer together. The particles eventually clump together to form a liquid.
Sublimation (solid to gas)
The particles gain kinetic energy and vibrate faster and faster. Eventually the forces of attraction between the particles are completely broken and they are able to escape from the solid.
Melting (solid to liquid)
The particles gain kinetic energy and vibrate faster and faster. This allows the particles to overcome the forces of attraction that hold them together in the solid. The regular pattern is broken down and the particles can now slide past one another.
Freezing (liquid to solid)
The particles lose kinetic energy and this allows the forces of attraction between the particles to hold them together. The particles arrange themselves into a regular pattern and are no longer able to slide past one another.
Boiling (liquid to gas)
The particles gain kinetic energy and move further apart. Eventually the forces of attraction between the particles are completely broken and they are able to escape from the liquid.
Condensation (gas to liquid)
The particles lose kinetic energy and this allows the forces of attraction to tiring the particles closer together. The particles eventually clump together to form a liquid.
Sublimation (solid to gas)
The particles gain kinetic energy and vibrate faster and faster. Eventually the forces of attraction between the particles are completely broken and they are able to escape from the solid.
b) Atoms
1.4: Describe and explain experiments to investigate the small size of particles and their movement
Dilution of colored solutions
Diffusion
Dilution of colored solutions
- Obtain a colored solution, such as water with food coloring added
- Add more water to the solution and observe the colors dilute
Diffusion
- Obtain a beaker of water
- Gradually add small drops of food coloring and watch them diffuse throughout the water
1.5: Understand the terms atom and molecule
- An atom is the basic unit of a chemical element and is made up of subatomic particles called protons, neutrons and electrons
- Most molecules are made up of two or more atoms covalently bonded together. However, noble gases exist as atoms at room temperature so their atom and molecule are the same.
1.6: Understand the differences between elements, compounds and mixtures
- Elements are substances that cannot be broken down by chemical means
- Compounds are substances that are made up of at least two elements that are chemically bonded together
- Mixtures are made up of elements or compounds that are not chemically bonded
1.7: Describe experimental techniques for the separation of mixtures
Simple Distillation (to separate a liquid from a solution)
Fractional Distillation (to separate two or more liquids that are miscible with one another)
Filtration (to separate an undissolved solid from a mixture of the solid and a liquid/solution)
Crystallization (to separate a dissolved solid from a solution, when the solid is much more soluble in hot solvent than in cold)
Paper Chromatography (to separate substances that have different solubilities in a given solvent)
Simple Distillation (to separate a liquid from a solution)
- Heat the solution of two liquids to the boiling point of the solvent
- The solvent will evaporate and its vapor will be cooled in the condenser
- The liquid distillate can then be collected
Fractional Distillation (to separate two or more liquids that are miscible with one another)
- Heat a water and ethanol solution to 78°C (the boiling point of ethanol)
- The ethanol will evaporate, cool then condense before the water does, as the boiling point of water is higher
- The fractional distillation of crude oil can be used as an alternative example
Filtration (to separate an undissolved solid from a mixture of the solid and a liquid/solution)
- Obtain an insoluble solid such as sand and place it in a beaker of water
- Pour the contents of the beaker into filter paper
- The sand will rest on the filter paper and the water will flow into the beaker below
Crystallization (to separate a dissolved solid from a solution, when the solid is much more soluble in hot solvent than in cold)
- Heat copper sulphate crystals that have been dissolved in copper sulphate solution
- The copper sulphate will evaporate and copper sulphate crystals will be left in the evaporating basin
Paper Chromatography (to separate substances that have different solubilities in a given solvent)
- A baseline is drawn in pencil on the chromatography paper. Ink must not be used, as the colors in the ink might separate and move up the paper.
- A small spot of the unknown mixture is placed alongside spots of pure substances which may be present.
- The bottom edge of the paper is then dipped in the solvent (usually water) and left while the solvent slowly soaks up to the top of the paper
- The solvent level must start below the baseline so that the substances being separated don't dissolve off the paper into the bulk of the solvent
1.8: Explain how information from chromatograms can be used to identify the composition of a mixture
Paper chromatography is used to separate substances that have different solubilities in a given solvent. Chromatography paper is placed in a solvent, the different compounds will travel at different speeds (due to the size of their particles). The shape on the chromatogram can be compared with that of known substances and where they match they are the same substance.
Paper chromatography is used to separate substances that have different solubilities in a given solvent. Chromatography paper is placed in a solvent, the different compounds will travel at different speeds (due to the size of their particles). The shape on the chromatogram can be compared with that of known substances and where they match they are the same substance.
c) Atomic structure
|
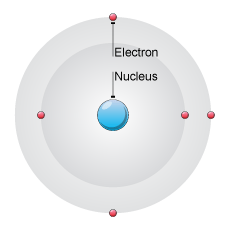
1.9: Understand that atoms consist of a central nucleus, composed of protons and neutrons, surrounded by electrons, orbiting in shells
Atoms consist of a nucleus in the center. The nucleus contains positively charged protons and neutral neutrons. The electrons that orbit the nucleus in shells are negatively charged and they are much smaller than protons and neutrons. |
1.11: Understand the terms atomic number, mass number, isotopes, and relative atomic mass
- The atomic number is the number of protons in the nucleus of the atom
- The mass number is the number of protons and neutrons in the nucleus of the atom
- An isotope is an atom with a different number of neutrons than protons in the nucleus
1.12: Calculate the relative atomic mass of an element from the relative abundance of its isotopes
Worked Example
Chlorine has two isotopes: chlorine-35 and chlorine-37
A typical sample of chlorine will be 75% chlorine-35 atoms and 25% chlorine-37 atoms.
The relative atomic mass is calculated as follows:
Total mass of 100 atoms = (75 x 35) + (25 x 37) = 3550
Mean mass of one atom = (3550 ÷ 100) = 35.5
Ar of chlorine is 35.5
Worked Example
Chlorine has two isotopes: chlorine-35 and chlorine-37
A typical sample of chlorine will be 75% chlorine-35 atoms and 25% chlorine-37 atoms.
The relative atomic mass is calculated as follows:
Total mass of 100 atoms = (75 x 35) + (25 x 37) = 3550
Mean mass of one atom = (3550 ÷ 100) = 35.5
Ar of chlorine is 35.5
1.13: Understand that the Periodic Table is an arrangement of elements in order to atomic number
The Periodic Table includes all of the elements and they are arranged in order of atomic number. The atomic number of the element is the number of protons in an atom. The number of protons in an atom is the same as the number of electrons.
The Periodic Table includes all of the elements and they are arranged in order of atomic number. The atomic number of the element is the number of protons in an atom. The number of protons in an atom is the same as the number of electrons.
1.14: Deduce the electronic configurations of the first 20 elements from their positions in the Periodic Table
- Hydrogen - 1
- Helium - 2
- Lithium - 2, 1
- Beryllium - 2, 2
- Boron - 2, 3
- Carbon - 2, 4
- Nitrogen - 2, 5
- Oxygen - 2, 6
- Fluorine - 2, 7
- Neon - 2, 8
- Sodium - 2, 8, 1
- Magnesium - 2, 8, 2
- Aluminium - 2, 8, 3
- Silicon - 2, 8, 4
- Phosphorus - 2, 8, 5
- Sulfur - 2, 8, 6
- Chlorine - 2, 8, 7
- Argon - 2, 8, 8
- Potassium - 2, 8, 8, 1
- Calcium - 2, 8, 8, 2
1.15: Deduce the number of outer electrons in a main group element from its position in the Periodic Table
The number of outer electrons in an element can be deduced by looking at the element's group number. Groups in the Periodic Table go across and they can be used to determine the number of valence electrons that an element has. For example, lithium, in group one, has one valence electron. Neon in group eight has eight valence electrons.
The number of outer electrons in an element can be deduced by looking at the element's group number. Groups in the Periodic Table go across and they can be used to determine the number of valence electrons that an element has. For example, lithium, in group one, has one valence electron. Neon in group eight has eight valence electrons.
d) Relative formula masses and molar volumes of gases
1.16: Calculate relative formula masses from relative atomic masses
In order to calculate the Mr of a substance, you need to simply add up the relative atomic masses of all the atoms present in the formula.
In order to calculate the Mr of a substance, you need to simply add up the relative atomic masses of all the atoms present in the formula.
1.17: Understand the use of the term mole to represent the amount of substance
The mole is the measure of the amount of substance. For example, one mol of sodium contains 6.02 x 1023 atoms of sodium.1.18: Understand the term mole as the Avogadro number of particles in a substance
The Avogadro number is the number of particles in a substance. 6.02 x 1023 is the Avogadro number.1.19: Carry out mole calculations using relative atomic mass and relative formula mass
The mass of one mole of atoms can easily be calculated. It is simply the relative atomic mass (Ar) expressed in grams.
The mass of one mole of a substance can also easily be calculated. It is simply the formula mass (Mr) expressed in grams.
The mass of one mole of atoms can easily be calculated. It is simply the relative atomic mass (Ar) expressed in grams.
The mass of one mole of a substance can also easily be calculated. It is simply the formula mass (Mr) expressed in grams.
1.20: Understand the the term molar volume of a gas and use its values at room temperature and pressure in calculations
One mole of any gas has a volume of 24dm3 at room temperature and pressure (rtp). This is called the molar volume of a gas.
Volume of gas (in dm3) = amount x 24
e) Chemical formulae and chemical equations
1.21: Write word equations and balanced chemical equations to represent chemical reactions
Word equations are equations that show chemical reactions using the names of the reactants and products involved.
Balanced chemical equations are equations that show chemical reactions using the symbols of the reactants and products involved. The symbols include the numbers of each element or compound. There must be an equal number of each element on each side of the equation, otherwise it must be balanced.
Word equations are equations that show chemical reactions using the names of the reactants and products involved.
Balanced chemical equations are equations that show chemical reactions using the symbols of the reactants and products involved. The symbols include the numbers of each element or compound. There must be an equal number of each element on each side of the equation, otherwise it must be balanced.
1.22: Use the state symbols (s), (l), (g) and (aq) in chemical equations to represent solids, liquids, gases and aqueous solutions respectively
In balanced chemical equations, the state symbols (s), (l), (g), and (aq) are written after an element or compound to show what state it is in. Solids are expressed using (s). Liquids, mainly water, are expressed using (l). Gases are expressed using (g), and aqueous solutions are expressed using (aq).
In balanced chemical equations, the state symbols (s), (l), (g), and (aq) are written after an element or compound to show what state it is in. Solids are expressed using (s). Liquids, mainly water, are expressed using (l). Gases are expressed using (g), and aqueous solutions are expressed using (aq).
1.23: Understand how the formulae of simple compounds can be obtained experimentally, including metal oxides, water and salts containing water of crystallization
The chemical formulae of chemical compounds can be obtained by conducting experiments with the compounds to determine how much there is of each element in a compound. There are two common experimental methods which help obtain this information. The method used depends on the type of compounds.
The metal oxide method involves heating a known mass of magnesium strongly. During heating, the magnesium reacts with oxygen in the air and its mass increases when it changes to magnesium oxide. The increase in mass needs to be measured to calculate the amount of oxygen which has reacted with the magnesium during the oxidation reaction. The empirical formula can then be calculated.
The hydrated salts method involves heating a hydrated salt until it decomposes and the water of crystallization from the compound evaporates. The mass of the salt decreases because of the lost water. The compound is heated until the mass does not decrease any further, and the decrease in mass can be used to calculate the mass of water lost.
The chemical formulae of chemical compounds can be obtained by conducting experiments with the compounds to determine how much there is of each element in a compound. There are two common experimental methods which help obtain this information. The method used depends on the type of compounds.
The metal oxide method involves heating a known mass of magnesium strongly. During heating, the magnesium reacts with oxygen in the air and its mass increases when it changes to magnesium oxide. The increase in mass needs to be measured to calculate the amount of oxygen which has reacted with the magnesium during the oxidation reaction. The empirical formula can then be calculated.
The hydrated salts method involves heating a hydrated salt until it decomposes and the water of crystallization from the compound evaporates. The mass of the salt decreases because of the lost water. The compound is heated until the mass does not decrease any further, and the decrease in mass can be used to calculate the mass of water lost.
1.24: Calculate empirical and molecular formulae from experimental data
The empirical formula shows the simplest whole number molar ratio of the atoms/ions in a unit and can be calculated from experimental results, using moles. The empirical formula can also be used to indicate the ratio of salt particle to water molecules in a hydrated salt. Sometimes, the empirical formula can be derived without using a calculation (e.g. C6H6 = CH)
However, sometimes experimental data must be used to calculate the empirical formula, using moles:
The empirical formula shows the simplest whole number molar ratio of the atoms/ions in a unit and can be calculated from experimental results, using moles. The empirical formula can also be used to indicate the ratio of salt particle to water molecules in a hydrated salt. Sometimes, the empirical formula can be derived without using a calculation (e.g. C6H6 = CH)
However, sometimes experimental data must be used to calculate the empirical formula, using moles:
|
mass (g) molar mass number of moles molar ratio |
carbon
48g 12 4 4 ÷ 4 = 1 |
hydrogen
16g 1 16 16 ÷ 4 = 4 |
In the above example, there is one atom of carbon for every four atoms of hydrogen. This makes the empirical formula CH4. The data can also be represented in percentage form:
|
mass (g) molar mass number of moles molar ratio |
carbon
75% 12 6.25 6.25 ÷ 6.25 = 1 |
hydrogen
25% 1 25 25 ÷ 6.25 = 4 |
The molecular formula shows the actual number of atoms in a compound and is simply a multiple of the empirical formula. However, there are many possible molecular formulae for one particular empirical formula. If the molar mass of the substance is known, the molecular formula can be calculated using the following method:
- Calculate the mass of the empirical formula [84g of CH2 = (12g x 1) + (1g x 2) = 14g]
- Divide the molar mass by the empirical mass to find n [84 ÷ 14 = 6]
- Multiply empirical formula by n to find the molecular formula [CH2 x 6 = C6H12]
1.25: Calculate reacting masses using experimental data and chemical equations
Balanced equations and simple molar calculations can be used to predict the masses of reactants and products. For example:
Balanced equations and simple molar calculations can be used to predict the masses of reactants and products. For example:
|
mass molar mass moles |
CaCO3
25g 100 0.25 |
CaO
14g 56 0.25 |
CO2
11g 44 0.25 |
1.26: Calculate percentage yield
The yield of a reaction is the mass of the product it produces. Percentage yield tells you about the overall success of an experiment. It compares what you think you should get (i.e. the theoretical yield), with what you actually get - the actual yield. The percentage yield is expressed as a percentage. Percentage yield can be calculated using the following formula:
percentage yield = (obtained yield ÷ theoretical yield) x 100
Yields are always less than 100% because in real life, some product or reactant always gets lost during a reaction. This is due to:
Chemists in industry are always looking for ways to produce products in a way that safely makes the most profit (i.e. the highest possible percentage yield).
The yield of a reaction is the mass of the product it produces. Percentage yield tells you about the overall success of an experiment. It compares what you think you should get (i.e. the theoretical yield), with what you actually get - the actual yield. The percentage yield is expressed as a percentage. Percentage yield can be calculated using the following formula:
percentage yield = (obtained yield ÷ theoretical yield) x 100
Yields are always less than 100% because in real life, some product or reactant always gets lost during a reaction. This is due to:
- Incomplete reactions - not all of the reactant is converted to product
- Practical losses during preparation - you might lose some reactant or product when you transfer chemicals from one container to another
- Unwanted reactions - these can be caused by impurities in the reactants
Chemists in industry are always looking for ways to produce products in a way that safely makes the most profit (i.e. the highest possible percentage yield).
1.27: Carry out mole calculations using volumes and molar concentrations
Bottles of solution in the lab are labelled to show the concentration of the solution. The concentration is sometimes called the molarity of the solution. The concentration 1M means that there is one mole of the substance dissolved in one decimeter cubed of its solution. The concentration of a solution can be calculated using the following formula:
concentration (mol/dm³) = number of moles ÷ volume in dm³
Bottles of solution in the lab are labelled to show the concentration of the solution. The concentration is sometimes called the molarity of the solution. The concentration 1M means that there is one mole of the substance dissolved in one decimeter cubed of its solution. The concentration of a solution can be calculated using the following formula:
concentration (mol/dm³) = number of moles ÷ volume in dm³
f) Ionic compounds
1.28: Describe the formation of ions by the gain or loss of electrons
An ion is an electrically charged atom or group of atoms. Ions are formed by the loss or gain of electrons.
An ion is an electrically charged atom or group of atoms. Ions are formed by the loss or gain of electrons.
1.29: Understand oxidation as the loss of electrons and reduction as the gain of electrons
Oxidation is the loss of electrons from an atom. When electrons are lost, the atom becomes positively charged.
Reduction is the gain of electrons from an atom. When electrons are gained, the atom becomes negatively charged.
Oxidation is the loss of electrons from an atom. When electrons are lost, the atom becomes positively charged.
Reduction is the gain of electrons from an atom. When electrons are gained, the atom becomes negatively charged.
1.30: Recall the charges of common ions
1.31: Deduce the charge of an ion from the electronic configuration of the atom from which the ion is formed
In the Periodic Table, each group represents how many valence electrons each atom contains. Using these clues, you can find out the charge of an ion of each element.
Group 1: 1+ charge
Group 2: 2+ charge
Group 3: Varying amounts of charge
Group 4: Varying amounts of charge
Group 5: 3- charge
Group 6: 2- charge
Group 7: 1- charge
In the Periodic Table, each group represents how many valence electrons each atom contains. Using these clues, you can find out the charge of an ion of each element.
Group 1: 1+ charge
Group 2: 2+ charge
Group 3: Varying amounts of charge
Group 4: Varying amounts of charge
Group 5: 3- charge
Group 6: 2- charge
Group 7: 1- charge
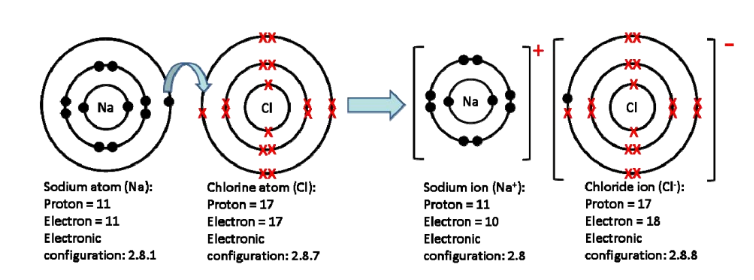
1.32: Explain, using dot and cross diagrams, the formation of ionic compounds by electron transfer, limited to elements from Groups 1, 2, 3 and 5, 6, 7
1.33: Understand ionic bonding as a strong electrostatic attraction between oppositely charged ions
Metallic elements normally lose electrons; nonmetals usually gain electrons. They exchange electrons so they will both reach stable electronic configurations. The resulting oppositely charged ions have strong electrostatic attraction between them; it is very hard to split ionically bonded atoms.
Metallic elements normally lose electrons; nonmetals usually gain electrons. They exchange electrons so they will both reach stable electronic configurations. The resulting oppositely charged ions have strong electrostatic attraction between them; it is very hard to split ionically bonded atoms.
1.34: Understand that ionic compounds have high melting and boiling points because of strong electrostatic forces between oppositely charged ions
Ionic bonds are very strong so a lot of energy is needed to break them. Ionic compounds contain many ionic bonds and therefore they have very high melting and boiling points.
Ionic bonds are very strong so a lot of energy is needed to break them. Ionic compounds contain many ionic bonds and therefore they have very high melting and boiling points.
1.35: Understand the relationship between ionic charge and the melting point and boiling point of an ionic compound
Ionic substances form giant ionic lattices. The oppositely charged ions have very strong forces of attraction between them and therefore lots of energy is required in order to break the bonds. This is why they have high melting and boiling points.
Ionic substances form giant ionic lattices. The oppositely charged ions have very strong forces of attraction between them and therefore lots of energy is required in order to break the bonds. This is why they have high melting and boiling points.
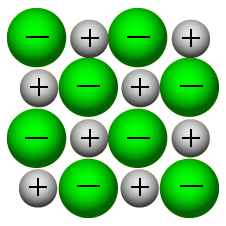
1.36: Describe an ionic crystal as a giant three-dimensional lattice structure held together by the attraction between oppositely charged ions
Ionic substances form giant three-dimensional lattices with very strong forces of attraction between the oppositely charged ions contained within the structure.
Ionic substances form giant three-dimensional lattices with very strong forces of attraction between the oppositely charged ions contained within the structure.
1.37: Draw a diagram to represent the positions of the ions in a crystal of sodium chloride
g) Covalent substances
1.38: Describe the formation of a covalent bond by the sharing of a pair of electrons between two atoms
A covalent bond is where two non-metallic atoms share electrons so that they can both obtain a full outer electron shell of either eight or two.
A covalent bond is where two non-metallic atoms share electrons so that they can both obtain a full outer electron shell of either eight or two.
1.39: Understand covalent bonding as a strong attraction between the bonding pair of electrons and the nuclei of the atoms involved in the bond
The electrons of the atoms that are involved in the bond are strongly attracted to the nuclei of the atoms.
The electrons of the atoms that are involved in the bond are strongly attracted to the nuclei of the atoms.
1.40: Explain, using dot and cross diagrams, the formation of covalent compounds by electron sharing for the following substances
You need to know how to draw the dot and cross diagrams for the following substances and explain how they form covalent compounds:
You need to know how to draw the dot and cross diagrams for the following substances and explain how they form covalent compounds:
- hydrogen
- chlorine
- hydrogen chloride
- water
- methane
- ammonia
- oxygen
- nitrogen
- carbon dioxide
- ethane
- ethene
1.41: Understand that substances with simple molecular structures are gases or liquids, or solids with low melting points
Substance with simple molecular structures are usually gases or liquids. This is because the intermolecular forces are not very strong and therefore the particles can move more freely than if the substance was a solid. If the substance is a solid, it usually has a low melting point because the weak bonds can be broken easily.
Substance with simple molecular structures are usually gases or liquids. This is because the intermolecular forces are not very strong and therefore the particles can move more freely than if the substance was a solid. If the substance is a solid, it usually has a low melting point because the weak bonds can be broken easily.
1.42: Explain why substances with simple molecular structures have low melting and boiling points in terms of the relatively weak forces between the molecules
Substances with simple molecular structures have low melting and boiling points because they have relatively weak forces binding the molecules together. Within the molecules themselves, the covalent bonds are very strong, but the intermolecular forces are weak. Because of this, the melting and boiling points are low since the molecules can be broken apart easily.
Substances with simple molecular structures have low melting and boiling points because they have relatively weak forces binding the molecules together. Within the molecules themselves, the covalent bonds are very strong, but the intermolecular forces are weak. Because of this, the melting and boiling points are low since the molecules can be broken apart easily.
1.43: Explain the high melting and boiling points of substances with giant covalent structures in terms of the breaking of many strong covalent bonds
The intramolecular bonds in giant covalent substances are very strong. Lots of heat energy is required to break these strong covalent bonds is required, which is why giant covalent substances have high melting and boiling points.
The intramolecular bonds in giant covalent substances are very strong. Lots of heat energy is required to break these strong covalent bonds is required, which is why giant covalent substances have high melting and boiling points.
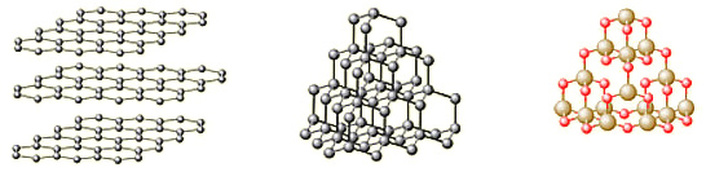
1.44: Draw diagrams representing the positions of the atoms in diamond and graphite
1.45: Explain how the uses of diamond and graphite depend on their structures, limited to graphite as a lubricant and diamond in cutting
Atoms in graphite are in a form where they are in layers. These layers can slide over one another, so graphite is much softer than diamond. Graphite is used as a lubricant because of these properties. For example, graphite is used in pencil - when you are writing, the layers of graphite are sliding off the tip of the pencil and onto the paper.
Unlike graphite, diamond is very hard, and this is why it is used for cutting. Each carbon atom in diamond is covalently bonded to four other carbon atoms, giving each atom a full valence shell and resulting in very strong intramolecular bonds.
Atoms in graphite are in a form where they are in layers. These layers can slide over one another, so graphite is much softer than diamond. Graphite is used as a lubricant because of these properties. For example, graphite is used in pencil - when you are writing, the layers of graphite are sliding off the tip of the pencil and onto the paper.
Unlike graphite, diamond is very hard, and this is why it is used for cutting. Each carbon atom in diamond is covalently bonded to four other carbon atoms, giving each atom a full valence shell and resulting in very strong intramolecular bonds.
h) Metallic crystals
1.46: Understand that a metal can be described as a giant structure of positive ions surrounded by a sea of delocalized electrons
When metals ionically bond with non-metals, they usually lose electrons which makes them positively charged cations. Some metals have many electron shells, since the metal ions are so large. The electrons that are furthest away from the nucleus of the ion have very weak forces of attraction binding them to the nucleus, so they are able to leave one ion and jump to the outermost shells of the next one.
When metals ionically bond with non-metals, they usually lose electrons which makes them positively charged cations. Some metals have many electron shells, since the metal ions are so large. The electrons that are furthest away from the nucleus of the ion have very weak forces of attraction binding them to the nucleus, so they are able to leave one ion and jump to the outermost shells of the next one.
1.47: Explain the electrical conductivity and malleability of a metal in terms of its structure and bonding
Metals can conduct electricity because when they ionically bond, they lose electrons in order to balance the number of protons and electrons in the nuclei of their atoms. In a metal, the electrons surrounding the positively charged nuclei can jump from atom to atom. The moving of electrons creates electric current: the basis of electricity.
Metals can conduct electricity because when they ionically bond, they lose electrons in order to balance the number of protons and electrons in the nuclei of their atoms. In a metal, the electrons surrounding the positively charged nuclei can jump from atom to atom. The moving of electrons creates electric current: the basis of electricity.
i) Electrolysis
1.48: Understand an electric current as a flow of electrons or ions
An electric current is a flow of electrons. For a substance to conduct electricity it must have mobile charged particles, which can either be electrons or ions. For instance, metals can conduct electricity because they have delocalized electrons which flow to create an electric current.
An electric current is a flow of electrons. For a substance to conduct electricity it must have mobile charged particles, which can either be electrons or ions. For instance, metals can conduct electricity because they have delocalized electrons which flow to create an electric current.
1.49: Understand why covalent compounds do not conduct electricity
Covalent compounds do not have any mobile charged particles. They consist of atoms or molecules which are electrically neutral. They cannot conduct electricity even when melted or dissolved in water.
Covalent compounds do not have any mobile charged particles. They consist of atoms or molecules which are electrically neutral. They cannot conduct electricity even when melted or dissolved in water.
1.50: Understand why ionic compounds conduct electricity only when molten or in solution
Ionic compounds have charged particles (metal cations and non-metal anions). However, ionic compounds can only conduct electricity when the ionic compound is molten or dissolved in water. Only in the liquid or aqueous state can the ions move to the oppositely charged electrode.
Ionic compounds have charged particles (metal cations and non-metal anions). However, ionic compounds can only conduct electricity when the ionic compound is molten or dissolved in water. Only in the liquid or aqueous state can the ions move to the oppositely charged electrode.
1.51: Describe experiments to distinguish between electrolytes and non-electrolytes
A liquid (molten or solution) that conducts electricity is called an electrolyte. Substances can be tested for if they are electrolytes or non-electrolytes with relatively simple experiments as described below:
A liquid (molten or solution) that conducts electricity is called an electrolyte. Substances can be tested for if they are electrolytes or non-electrolytes with relatively simple experiments as described below:
|
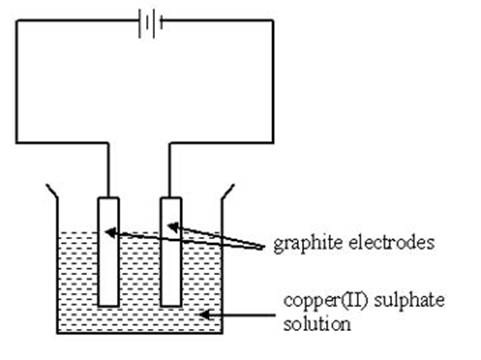
A basic electrolytic circuit is shown on the left. It consists of a power supply, wires and graphite electrodes. Graphite is a carbon compound that is chosen as a material for the electrodes because it is inert - it is chemically uncreative and will not interfere with the electrolysis reaction. The electrode which is connected to the positive terminal of the battery is called the anode; the one connected to the negative terminal is called the cathode. The substance to be tested in this case is copper(II) sulphate dissolved in solution. Molten substances can also be tested using this method. If an ammeter is connected to the circuit and current is detected, the substance tested is an electrolyte.
|
1.52: Understand that electrolysis involves the formation of new substances when ionic compounds conduct electricity
An electrolyte is a different type of conductor than a solid metal. When it conducts, the current passing through the electrolyte causes a chemical reaction to take place, which forms new substances. The ionic compound which makes up the electrolyte is decomposed. This reaction is called an electrolysis reaction; new substances are formed when the molten or aqueous ionic compound conducts electricity. The current causes a chemical reaction which converts electrical energy into chemical energy.
An electrolyte is a different type of conductor than a solid metal. When it conducts, the current passing through the electrolyte causes a chemical reaction to take place, which forms new substances. The ionic compound which makes up the electrolyte is decomposed. This reaction is called an electrolysis reaction; new substances are formed when the molten or aqueous ionic compound conducts electricity. The current causes a chemical reaction which converts electrical energy into chemical energy.
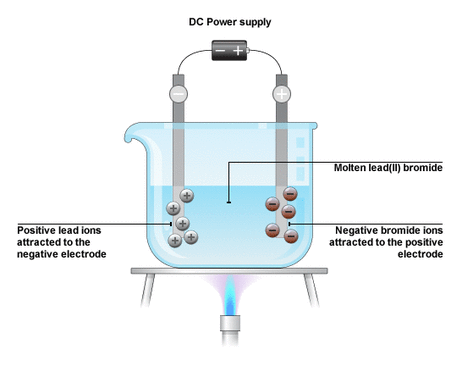
1.53: Describe simple experiments for the electrolysis, using inert electrodes, of molten salts such as lead(II) bromide
When a molten salt is electrolyzed, the positive metal ions are attracted to the negative electrode, or cathode. It is here where they lose their charge as they gain electrons and become metal atoms. The negative non-metal ions are attracted to the positive electrode, or anode. It is here where they lose their charge as they lose electrons and become non-metal atoms. They usually form molecules. An example, the electrolysis of lead(II) bromide, is shown below:
When a molten salt is electrolyzed, the positive metal ions are attracted to the negative electrode, or cathode. It is here where they lose their charge as they gain electrons and become metal atoms. The negative non-metal ions are attracted to the positive electrode, or anode. It is here where they lose their charge as they lose electrons and become non-metal atoms. They usually form molecules. An example, the electrolysis of lead(II) bromide, is shown below:
|
As you can see, the molten lead(II) bromide is decomposed and split into lead atoms and diatomic bromine molecules because of their conductivity. This suggests that when a molten salt is electrolyzed, the metal is produced at the cathode whilst the non-metal is formed at the anode.
Half-equation at the cathode Pb2+ + 2e- → Pb Half-equation at the anode 2Br- → Br2 + 2e- |
1.54: Describe simple experiments for the electrolysis, using inert electrodes, of aqueous solutions of sodium chloride, copper(II) sulphate and dilute sulphuric acid and predict the products
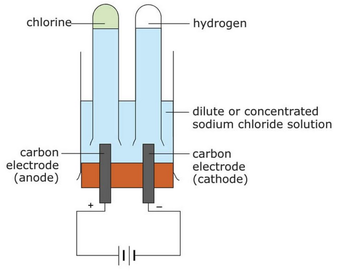
When a solution is electrolyzed, predicting the products becomes more complicated as the water (the solvent) also becomes involved. Even though water is a covalent compound, ionizes a little bit during electrolysis, producing hydrogen and hydroxide ions which are also attracted to the electrodes. In most cases, a gas will be produced which needs to be collected and tested. When carrying out electrolysis during which a gas will be produced, the following setup can be used to collect the gas:
When a solution is electrolyzed, predicting the products becomes more complicated as the water (the solvent) also becomes involved. Even though water is a covalent compound, ionizes a little bit during electrolysis, producing hydrogen and hydroxide ions which are also attracted to the electrodes. In most cases, a gas will be produced which needs to be collected and tested. When carrying out electrolysis during which a gas will be produced, the following setup can be used to collect the gas:
|
For any concentrated solution of a salt made up from a Group 1 metal and a Group 7 non-metal, there will always be three products: hydrogen, the halogen, and the hydroxide of the Group 1 metal.
Electrolysis of sodium chloride solution At the anode, yellow-green chlorine gas will be produced in a nearly 1:1 ratio with the gas produced at the cathode. This gas bleaches damp litmus paper. At the cathode, colorless hydrogen gas will be produced. The solution around the cathode will bubble. There are two tests for hydrogen gas: it will turn red litmus paper blue, and a lit splint will make a 'pop' sound. |
Electrolysis of copper(II) sulphate solution
At the anode, colorless hydroxide gas will be produced. The solution around the anode will bubble. The gas will relight a glowing splint.
At the cathode, copper is discharged instead of the hydrogen ions; they remain in the solution. There will be visible red deposits of copper on the electrode.
Electrolysis of dilute sulphuric acid
The electrolyte, sulphuric acid, is diluted and this has an effect on the kind of products of the electrolysis, as none of the ions of the electrolyte will be discharged. Instead, they will remain in the solution. The ions which are discharged are hydrogen ions at the cathode and hydroxide ions at the anode. This shows that when a dilute solution is electrolyzed, the water is decomposed rather than the electrolyte. The identity and nature of the electrolyte does not have an effect on the products in the electrolysis of dilute solutions.
At the anode, colorless hydroxide gas will be produced. The solution around the anode will bubble. The gas will relight a glowing splint.
At the cathode, copper is discharged instead of the hydrogen ions; they remain in the solution. There will be visible red deposits of copper on the electrode.
Electrolysis of dilute sulphuric acid
The electrolyte, sulphuric acid, is diluted and this has an effect on the kind of products of the electrolysis, as none of the ions of the electrolyte will be discharged. Instead, they will remain in the solution. The ions which are discharged are hydrogen ions at the cathode and hydroxide ions at the anode. This shows that when a dilute solution is electrolyzed, the water is decomposed rather than the electrolyte. The identity and nature of the electrolyte does not have an effect on the products in the electrolysis of dilute solutions.
1.55: Write ionic half-equations representing the reactions at the electrodes during electrolysis
At the positive electrode, electrons will be lost: to show this, the lost electrons are written as reactants. At the negative electrode, electrons will be gained: to show this, the gained electrons are written as products.
Cathode reaction during the electrolysis of dilute sulphuric acid (reduction)
2H+ + 2e- → H2
Anode reaction during the electrolysis of dilute sulphuric acid (oxidation)
4OH- → O2 + 2H2O + 4e-
At the positive electrode, electrons will be lost: to show this, the lost electrons are written as reactants. At the negative electrode, electrons will be gained: to show this, the gained electrons are written as products.
Cathode reaction during the electrolysis of dilute sulphuric acid (reduction)
2H+ + 2e- → H2
Anode reaction during the electrolysis of dilute sulphuric acid (oxidation)
4OH- → O2 + 2H2O + 4e-
1.56: Recall that one faraday represents one mole of electrons
The amount of product of an electrolysis reaction can be calculated and depends on the amount of electrical charge passed through the electrolytic circuit. The amount of electrical charge is expressed as a number of faraday:
1 faraday = the charge of 1 mole of electrons = 96500 coulombs
The amount of charge that passes in a circuit depends on the current (measured in amperes) of the circuit and the amount of time (measured in seconds) that the current is switched on. The number of faraday can be calculated by multiplying the number of amperes by seconds, which gives coulombs, then dividing the number of coulombs by 96500. The greater the charge of the ions in the reaction, the greater the number of electrons needed to balance the equation.
The amount of product of an electrolysis reaction can be calculated and depends on the amount of electrical charge passed through the electrolytic circuit. The amount of electrical charge is expressed as a number of faraday:
1 faraday = the charge of 1 mole of electrons = 96500 coulombs
The amount of charge that passes in a circuit depends on the current (measured in amperes) of the circuit and the amount of time (measured in seconds) that the current is switched on. The number of faraday can be calculated by multiplying the number of amperes by seconds, which gives coulombs, then dividing the number of coulombs by 96500. The greater the charge of the ions in the reaction, the greater the number of electrons needed to balance the equation.
1.57: Calculate the amounts of the products of the electrolysis of molten salts and aqueous solutions
A solution of copper(II) sulphate is electrolyzed. How much copper will be deposited at the cathode by a current of 2A flowing for 20 minutes?
A solution of copper(II) sulphate is electrolyzed. How much copper will be deposited at the cathode by a current of 2A flowing for 20 minutes?
- Calculate the amount of charge (measured in coulombs) by multiplying the current (measured in amperes) by the amount of time (measured in seconds)
- Calculate the number of faraday by dividing the amount of charge by 96500
- Use the ratio in the reaction's half-equation to calculate the mass deposited using mass, molar mass and moles