|
Section 2: Chemistry of the elements
|
a) The Periodic Table
|
2.1: Understand the terms group and period
A group is a column of elements which goes down the Periodic Table and shows the number of valence electrons that each atom of that element has. A period goes across the Periodic Table and shows the number of electron shells that an atom of that element has. |
2.2: Recall the positions of metals and non-metals in the Periodic Table
The metals are on the left side of the Periodic Table and the non-metals on the right; they are separated by a staircase-like line which starts above aluminum and goes down next to polonium.
The metals are on the left side of the Periodic Table and the non-metals on the right; they are separated by a staircase-like line which starts above aluminum and goes down next to polonium.
2.3: Explain the classification of elements as metals or non-metals on the basis of their electrical conductivity and the acid-base character of their oxides
|
Metal
Non-metal |
Usually conducts electricity
Usually does not conduct electricity |
Acidic oxides
Base oxides |
2.4: Understand why elements in the same group of the Periodic Table have similar chemical properties
The Periodic Table was made based on the chemical properties of elements. The elements in the same group have similar chemical properties because a group with one metal in it will have only metals in it, and the same goes for non-metals. They also have the same number of valence electrons which makes them equally likely to form ions.
The Periodic Table was made based on the chemical properties of elements. The elements in the same group have similar chemical properties because a group with one metal in it will have only metals in it, and the same goes for non-metals. They also have the same number of valence electrons which makes them equally likely to form ions.
2.5: Understand that the noble gases (Group 0) are a family of inert gases and explain their lack of reactivity in terms of their electronic configurations
Noble gases are unreactive because they have a full valence shell of eight. This means that they cannot form covalent compounds since they already have a full valence shell. They cannot ionically bond because they already have a stable configuration of eight valence electrons. They cannot take part in metallic bonding because they are non-metals.
Noble gases are unreactive because they have a full valence shell of eight. This means that they cannot form covalent compounds since they already have a full valence shell. They cannot ionically bond because they already have a stable configuration of eight valence electrons. They cannot take part in metallic bonding because they are non-metals.
b) Group 1 elements - lithium, sodium and potassium
2.6: Describe the reaction of Group 1 elements with water and understand that the reactions provide a basis for their recognition as a family of elements
|
Group 1 element
Lithium Sodium Potassium |
Reaction with water
• Floats on the surface • Fizzes gently • Gives off hydrogen gas • Forms a colorless solution of lithium oxide • Enough heat is given off for the sodium to melt • Travels around the water surface • Does not sink • Gives off a trail of sodium hydroxide • Colorless sodium hydroxide solution is formed • Similar reaction to that of lithium • The hydrogen gas given off ignites with a lilac flame |
2.7: Describe the relative reactivities of the elements in Group 1
As you go down Group 1, the metals get more and more reactive (lithium simply fizzes in water, rubidium can blow up a bathtub)
As you go down Group 1, the metals get more and more reactive (lithium simply fizzes in water, rubidium can blow up a bathtub)
2.8: Explain the relative reactivities of the elements in Group 1 in terms of distance between the outer electrons and the nucleus
Since the bottom element in Group 1 has the most electrons, it has many shells that are very far from the nucleus. This means that the intermolecular forces are quite weak between the outer electrons and the nucleus so the electrons can ionically bond easily.
Since the bottom element in Group 1 has the most electrons, it has many shells that are very far from the nucleus. This means that the intermolecular forces are quite weak between the outer electrons and the nucleus so the electrons can ionically bond easily.
c) Group 7 elements - chlorine, bromine and iodine
2.9: Recall the colors and physical states of chlorine, bromine and iodine at room temperature
|
Element
Chlorine Bromine Iodine |
State at room temperature
Gas Liquid Solid |
Color at room temperature
Green Yellow/Brown Brown |
2.10: Make predictions about the properties of other halogens in Group 7
|
Element
Fluorine Astatine |
State at room temperature
Gas Solid |
Color at room temperature
Yellow Black |
2.11: Understand the difference between hydrogen chloride gas and hydrochloric acid
Hydrogen chloride gas is the same as hydrochloric acid except it is in a gaseous state; hydrochloric acid is an aqueous solution of hydrogen chloride
Hydrogen chloride gas is the same as hydrochloric acid except it is in a gaseous state; hydrochloric acid is an aqueous solution of hydrogen chloride
2.12: Explain, in terms of dissociation, why hydrogen chloride is acidic in water but not in methylbenzene
Hydrogen chloride reacts in water due to the presence of positive hydrogen ions in water. There are no hydrogen cations present in methylbenzene, so no reaction takes place.
Hydrogen chloride reacts in water due to the presence of positive hydrogen ions in water. There are no hydrogen cations present in methylbenzene, so no reaction takes place.
2.13: Describe the relative reactivities of the elements in Group 7
|
Element
Fluorine Chlorine Bromine Iodine Astatine |
Reactivity
Most reactive Reactivity of halogen decreases Reactivity of halogen decreases Reactivity of halogen decreases Least reactive |
2.14: Describe experiments to demonstrate that a more reactive halogen will displace a less reactive halogen from a solution of one of its salts
- Add chlorine water to potassium chloride, potassium bromide, and potassium iodide solutions
- Repeat the reactions with bromine water and iodine water
- Record the color changes
|
Reactant
Potassium Chloride Potassium Bromide Potassium Iodide |
Chlorine water
No reaction Turns orange Turns brown |
Bromine water
No reaction No reaction Turns brown |
Iodine water
No reaction No reaction No reaction |
2.15: Understand these displacement reactions as redox reactions
These displacement reactions are known as redox reactions since both oxidation and reduction take place.
These displacement reactions are known as redox reactions since both oxidation and reduction take place.
d) Oxygen and oxides
2.16: Recall the gases present in air and their approximate percentage by volume
- Nitrogen (N2) makes up about 78% of air
- Oxygen (O2) makes up about 21% of air
- Carbon dioxide (CO2) makes up about 0.03% of air
- Other gases like argon make up about 1% of air
2.17: Explain how experiments involving the reactions of elements such as copper, iron and phosphorus with air can be used to determine the percentage by volume of oxygen in air
The percentage of oxygen in the air can be measured by passing a known volume of air over hot copper, and measuring the decrease in volume as the oxygen reacts with it. Gas syringes are used to measure the volume of gas in the experiment. Note that there is some air in the tube with the copper turnings. The oxygen in this air will also react with the hot copper, causing a small error in the final volume recorded. It is also important to let the apparatus cool down at the end of the experiment, otherwise the final reading will be too high.
The percentage of oxygen in the air can be measured by placing a piece of iron wool in a test tube which is inverted in a beaker of water. The test tube is filled with air and is left for a few days to allow the iron to rust. The length of the column of air in the test tube is measured at the start of the experiment and again when no more change to the length of the column or the iron wool is observed.
The percentage of oxygen in the air can be measured using phosphorus. A marked bell jar with its lid removed is placed on top of coins inside a trough. The coins ensure that water can go inside the bell jar. Water is poured into the trough until it reaches level zero marked on the bell jar. A piece of phosphorus is placed in an evaporating dish which floats on the water. The phosphorus is lit and a lid placed at the top of the bell jar to cover it. The yellow phosphorus starts burning, and the air space inside gets filled with white fumes. The white fumes are phosphorus oxide which dissolves in the water, forming an acidic solution. When the phosphorus has stopped burning and the white fumes have disappeared. The level of water in the bell jar is measured.
The percentage of oxygen in the air can be measured by passing a known volume of air over hot copper, and measuring the decrease in volume as the oxygen reacts with it. Gas syringes are used to measure the volume of gas in the experiment. Note that there is some air in the tube with the copper turnings. The oxygen in this air will also react with the hot copper, causing a small error in the final volume recorded. It is also important to let the apparatus cool down at the end of the experiment, otherwise the final reading will be too high.
The percentage of oxygen in the air can be measured by placing a piece of iron wool in a test tube which is inverted in a beaker of water. The test tube is filled with air and is left for a few days to allow the iron to rust. The length of the column of air in the test tube is measured at the start of the experiment and again when no more change to the length of the column or the iron wool is observed.
The percentage of oxygen in the air can be measured using phosphorus. A marked bell jar with its lid removed is placed on top of coins inside a trough. The coins ensure that water can go inside the bell jar. Water is poured into the trough until it reaches level zero marked on the bell jar. A piece of phosphorus is placed in an evaporating dish which floats on the water. The phosphorus is lit and a lid placed at the top of the bell jar to cover it. The yellow phosphorus starts burning, and the air space inside gets filled with white fumes. The white fumes are phosphorus oxide which dissolves in the water, forming an acidic solution. When the phosphorus has stopped burning and the white fumes have disappeared. The level of water in the bell jar is measured.
2.18: Describe the laboratory preparation of oxygen from hydrogen peroxide using manganese(II) oxide as a catalyst
In the laboratory, oxygen can be prepared using hydrogen peroxide, which spontaneously decomposes into water and oxygen as shown by the following equation:
At room temperature, this reaction is very slow and is therefore sped up using a catalyst such as manganese oxide. This reaction is called a catalytic decomposition. The catalyst and hydrogen peroxide are added to a flask; the oxygen gas is collected in a gas syringe.
In the laboratory, oxygen can be prepared using hydrogen peroxide, which spontaneously decomposes into water and oxygen as shown by the following equation:
- 2H2O2 → 2H2O + O2
At room temperature, this reaction is very slow and is therefore sped up using a catalyst such as manganese oxide. This reaction is called a catalytic decomposition. The catalyst and hydrogen peroxide are added to a flask; the oxygen gas is collected in a gas syringe.
2.19: Describe the reactions with oxygen in air of magnesium, carbon and sulfur, and the acid-base character of the oxides produced
Oxygen produces the following oxides during combustion reactions:
Oxygen produces the following oxides during combustion reactions:
- Magnesium - white bright light, forming a white solid called magnesium oxide
- Carbon - colorless and odorless gas, carbon dioxide, produced
- Sulfur - pungent yellowish gas, sulfur dioxide, produced
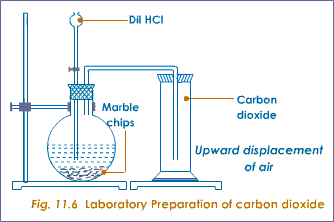
2.20: Describe the laboratory preparation of carbon dioxide from calcium carbonate and dilute hydrochloric acid
Carbon dioxide can be prepared using calcium carbonate (marble chips) and dilute hydrochloric acid:
Carbon dioxide can be prepared using calcium carbonate (marble chips) and dilute hydrochloric acid:
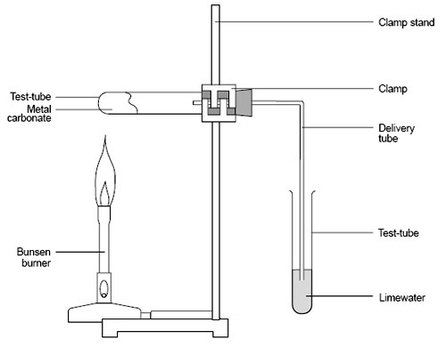
2.21: Describe the formation of carbon dioxide from the thermal decomposition of metal carbonates such as copper(II) carbonate
When copper(II) carbonate is heated it quickly decomposes into copper oxide and carbon dioxide:
When copper(II) carbonate is heated it quickly decomposes into copper oxide and carbon dioxide:
2.22: Recall the properties of carbon dioxide, limited to its solubility and density
Carbon dioxide is more dense than air and soluble in water at high pressures.
Carbon dioxide is more dense than air and soluble in water at high pressures.
2.23: Explain the uses of carbon dioxide in carbonating drinks and in fire extinguishers, in terms of its solubility and density
Carbon dioxide is denser than air so it smothers a fire preventing oxygen from getting to it. This is why it is used in fire extinguishers. It is also used in carbonating drinks because a large amount of carbon dioxide can be dissolved in water. The carbon dioxide is released when the bottle is opened and the pressure reduced. The fizzing is carbon dioxide leaving the solution.
Carbon dioxide is denser than air so it smothers a fire preventing oxygen from getting to it. This is why it is used in fire extinguishers. It is also used in carbonating drinks because a large amount of carbon dioxide can be dissolved in water. The carbon dioxide is released when the bottle is opened and the pressure reduced. The fizzing is carbon dioxide leaving the solution.
2.24: Understand that carbon dioxide is a greenhouse gas and may contribute to global warming
The Earth reradiates some of the energy it receives from the sun. Carbon dioxide in the atmosphere absorbs some of that heat so that not all of the heat goes back to space; this keeps the Earth warm. Gases which absorb the heat that the Earth reradiates are called greenhouse gases. Because of deforestation and increased burning of fossil fuels, there is now more carbon dioxide in the atmosphere than ever before. Evidence strongly suggests that as a result of this the Earth is warming and this causes climate changes as some places on Earth are getting hotter, others are getting colder, drier, windier or experience more destructive hurricanes or more severe draught.
The Earth reradiates some of the energy it receives from the sun. Carbon dioxide in the atmosphere absorbs some of that heat so that not all of the heat goes back to space; this keeps the Earth warm. Gases which absorb the heat that the Earth reradiates are called greenhouse gases. Because of deforestation and increased burning of fossil fuels, there is now more carbon dioxide in the atmosphere than ever before. Evidence strongly suggests that as a result of this the Earth is warming and this causes climate changes as some places on Earth are getting hotter, others are getting colder, drier, windier or experience more destructive hurricanes or more severe draught.
e) Hydrogen and water
2.25: Describe the reactions of dilute hydrochloric and dilute sulfuric acids with magnesium, aluminum, zinc and iron
Hydrogen gas can be made by reacting a metal such as magnesium, aluminum, zinc and iron with acids such as hydrochloric acid or sulfuric acid. These are all exothermic reactions. The metal displaces the hydrogen to form the acid. The general word equation for this type of reaction is:
Hydrogen gas can be made by reacting a metal such as magnesium, aluminum, zinc and iron with acids such as hydrochloric acid or sulfuric acid. These are all exothermic reactions. The metal displaces the hydrogen to form the acid. The general word equation for this type of reaction is:
- metal + acid → salt + hydrogen
2.26: Describe the combustion of hydrogen
Hydrogen is a colorless, odorless gas and has the lowest density of all gases; it is also very flammable and burns with oxygen as shown in the equation below. This is a very exothermic reaction and explains why hydrogen can be used as a fuel. The added advantage of using hydrogen as a fuel is that it is pollution-free and the reaction produces a harmless product: water.
Hydrogen is a colorless, odorless gas and has the lowest density of all gases; it is also very flammable and burns with oxygen as shown in the equation below. This is a very exothermic reaction and explains why hydrogen can be used as a fuel. The added advantage of using hydrogen as a fuel is that it is pollution-free and the reaction produces a harmless product: water.
- 2H2 + O2 → 2H2O
2.27: Describe the use of anhydrous copper(II) sulfate in the chemical test for water
The chemical test for water involves the use of anhydrous copper sulfate. If a liquid is added to white anhydrous copper(II) sulfate which then changes to a blue color, the liquid added must be water or contains water. The general equation for this reaction is:
The chemical test for water involves the use of anhydrous copper sulfate. If a liquid is added to white anhydrous copper(II) sulfate which then changes to a blue color, the liquid added must be water or contains water. The general equation for this reaction is:
- white anhydrous copper sulfate + liquid → blue hydrated copper sulfate
2.28: Describe a physical test to show whether water is pure
The purity of water can be tested by using a simple physical test like measuring its boiling point. Water is the only liquid which boils at 100°C; impure water would boil at a higher temperature. You should not use pH 7 as an indicator for water as water is not the only clear liquid to have a pH 7.
The purity of water can be tested by using a simple physical test like measuring its boiling point. Water is the only liquid which boils at 100°C; impure water would boil at a higher temperature. You should not use pH 7 as an indicator for water as water is not the only clear liquid to have a pH 7.
f) Reactivity series
2.29: Understand that metals can be arranged in a reactivity series based on the reactions of the metals and their compounds
Metals are arranged in a reactivity series in order of their reactivity. Here is the reactivity series, from most reactive to least:
Metals are arranged in a reactivity series in order of their reactivity. Here is the reactivity series, from most reactive to least:
- Potassium (K)
- Sodium (Na)
- Lithium (Li)
- Calcium (Ca)
- Magnesium (Mg)
- Aluminum (Al)
- Zinc (Zn)
- Iron (Fe)
- Copper (Cu)
2.30: Describe how reactions with water and dilute acids can be used to deduce the following in order of reactivity: potassium, sodium, lithium, calcium, magnesium, zinc, iron and copper
Only reactive metals react with water and this reaction produces a hydroxide and hydrogen. The rate at which hydrogen is released can be used to compare reactivity. The metals that react with cold water are all the group 1 metals.
The metals that do not react with cold water can be tested with steam. Reactive metals react very vigorously with steam and therefore steam should only really be used with those metals that show a low rate of reaction or no reaction with cold water.
Most metals also react with acids. When they do, they produce a salt and hydrogen according to the general equation:
Only reactive metals react with water and this reaction produces a hydroxide and hydrogen. The rate at which hydrogen is released can be used to compare reactivity. The metals that react with cold water are all the group 1 metals.
The metals that do not react with cold water can be tested with steam. Reactive metals react very vigorously with steam and therefore steam should only really be used with those metals that show a low rate of reaction or no reaction with cold water.
Most metals also react with acids. When they do, they produce a salt and hydrogen according to the general equation:
- metal + hydrochloric acid → metal chloride + hydrogen
2.31: Deduce the position of a metal within the reactivity series using displacement
reactions between metals and their oxides, and between metals and their
salts in aqueous solutions
When a metal is pure and unreacted it exists in the form of atoms. When a metal reacts with a non-metal, the metal forms positive ions. Therefore metals in solutions of ionic compounds are in the form of positive ions. A displacement reaction is a reaction in which the atoms of a more reactive metal displace the ions of a less reactive metal from its compound to become atoms again.This is because the more reactive the metal, the easier it forms ions. The less reactive the metal, the greater its tendency to exist as atoms.
When a metal is pure and unreacted it exists in the form of atoms. When a metal reacts with a non-metal, the metal forms positive ions. Therefore metals in solutions of ionic compounds are in the form of positive ions. A displacement reaction is a reaction in which the atoms of a more reactive metal displace the ions of a less reactive metal from its compound to become atoms again.This is because the more reactive the metal, the easier it forms ions. The less reactive the metal, the greater its tendency to exist as atoms.
2.32: Understand oxidation and reduction as the addition and removal of oxygen respectively
Oxidation is a reaction during which a substance gains oxygen and reduction is a reaction during which a substance loses oxygen.
Oxidation is a reaction during which a substance gains oxygen and reduction is a reaction during which a substance loses oxygen.
2.33: Understand the terms: redox, oxidizing agent and reducing agent
A redox reaction is a reaction during which both an oxidation and reduction occur. Oxygen is lost by one reactant and taken by another reactant. A substance that causes another substance to lose oxygen and becomes itself oxidized is called a reducing agent. A substance that loses oxygen to another substance becomes reduced and causes the other substance to be oxidized; such a substance is called an oxidizing agent.
A redox reaction is a reaction during which both an oxidation and reduction occur. Oxygen is lost by one reactant and taken by another reactant. A substance that causes another substance to lose oxygen and becomes itself oxidized is called a reducing agent. A substance that loses oxygen to another substance becomes reduced and causes the other substance to be oxidized; such a substance is called an oxidizing agent.
2.34: Describe the conditions under which iron rusts
Rusting is a reaction between iron, oxygen and water, producing a brittle red-brown product which is iron(III) oxide which weakens any iron structure.
Rusting is a reaction between iron, oxygen and water, producing a brittle red-brown product which is iron(III) oxide which weakens any iron structure.
2.35: Describe how the rusting of iron may be prevented by grease, oil, paint, plastic and galvanizing
The prevention of rusting should focus on ensuring all three reactants do not come into contact with each other.
Painting the iron surface, applying oil and grease, and covering it in plastic will create a barrier between the iron and oxygen/water. However, when the barrier is broken, rusting occurs.
Galvanizing (covering steel with zinc) works because zinc is more reactive/better at losing electrons than iron so the zinc is oxidized by the oxygen and water instead of the iron. The zinc can be chipped or scratched and the iron will still not rust. However, you will need to keep checking if there is enough zinc left on the iron.
The prevention of rusting should focus on ensuring all three reactants do not come into contact with each other.
Painting the iron surface, applying oil and grease, and covering it in plastic will create a barrier between the iron and oxygen/water. However, when the barrier is broken, rusting occurs.
Galvanizing (covering steel with zinc) works because zinc is more reactive/better at losing electrons than iron so the zinc is oxidized by the oxygen and water instead of the iron. The zinc can be chipped or scratched and the iron will still not rust. However, you will need to keep checking if there is enough zinc left on the iron.
2.36: Understand the sacrificial protection of iron in terms of the reactivity series
Sacrificial protection is when iron is protected with a more reactive metal. The more reactive metal is oxidized by the oxygen and water instead of the iron. The more reactive metal can be chipped and the iron will still not rust.
Sacrificial protection is when iron is protected with a more reactive metal. The more reactive metal is oxidized by the oxygen and water instead of the iron. The more reactive metal can be chipped and the iron will still not rust.
g) Tests for ions and gases
2.38: Describe simple tests for the cations
Lithium, sodium, potassium and calcium(II) ions can be detected using flame tests. When the metals catch fire, their flames will burn different colors:
Ammonium ions can be detected using sodium hydroxide solution. Add aqueous sodium hydroxide and heat, and then test the gas given off with damp red litmus paper. The red litmus will turn blue indicating the presence of ammonia.
Aqueous sodium hydroxide can be added to copper(II), iron(II), and iron(III) ions to indicate their presence:
Lithium, sodium, potassium and calcium(II) ions can be detected using flame tests. When the metals catch fire, their flames will burn different colors:
- the flame for lithium will be red
- the flame for sodium will be orange
- the flame for potassium will be lilac
- the flame for calcium(II) will be orange-red
Ammonium ions can be detected using sodium hydroxide solution. Add aqueous sodium hydroxide and heat, and then test the gas given off with damp red litmus paper. The red litmus will turn blue indicating the presence of ammonia.
Aqueous sodium hydroxide can be added to copper(II), iron(II), and iron(III) ions to indicate their presence:
- when sodium hydroxide is added to copper(II) ions, a blue precipitate will form, which is soluble in excess
- when sodium hydroxide is added to iron(II) ions, a green precipitate will form, which is soluble in excess
- when sodium hydroxide is added to iron(III) ions, an orange-brown precipitate will form, which is soluble in excess
2.39: Describe simple tests for the anions
- when chlorine is acidified with dilute nitric acid and aqueous silver nitrate is added, a white precipitate is formed
- when bromine is acidified with dilute nitric acid and aqueous silver nitrate is added, a pale cream precipitate is formed
- when iodine is acidified with dilute nitric acid and aqueous silver nitrate is added, a yellow precipitate is formed
- when sulfate is acidified with hydrochloric acid and aqueous barium chloride is added, a white precipitate is formed
- when carbonate is acidified with dilute acid, carbon dioxide is produced and this can be tested for with limewater
2.40: Describe simple tests for the gases hydrogen, oxygen, carbon dioxide, ammonia and chlorine
- hydrogen can be tested for with a lighted splint; the result is a squeaky 'pop'
- oxygen can be tested for with a glowing splint; oxygen will relight the splint
- carbon dioxide can be bubbled through limewater, and the result will be a milky, white precipitate
- ammonia can be tested for with damp red litmus paper, which will turn blue
- chlorine can be tested for with damp litmus paper, which will adopt a bleached appearance