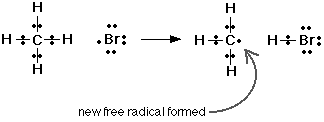|
Section 3: Organic chemistry
|
a) Introduction
|
3.1: Explain the terms homologous series, hydrocarbon, saturated, unsaturated, general formula and isomerism.
Homologous series A homologous series is a family of compounds with the same general formula and similar chemical properties. Hydrocarbon A hydrocarbon is a compound with only hydrogen and carbon atoms. Saturated Saturated means that there are the maximum number of carbon atoms possible without any double or triple bonds. Unsaturated Unsaturated means that the hydrocarbon contains double or triple bonds. General Formula CxHY is the general formula for a hydrocarbon. Isomerism Compounds with the same chemical formula but with a different displayed formula. |
b) Alkanes
3.2: Recall that alkanes have the general formula CnH2n+2
Alkanes have the general formula CnH2n+2
Alkanes have the general formula CnH2n+2
3.3: Draw displayed formulae for alkanes with up to five carbon atoms in a molecule, and name the straight-chain isomers
3.4: Recall the products of the complete and incomplete combustion of alkanes
Complete Combustion
Alkane + Oxygen → Carbon Dioxide + Water
Note: The complete combustion is when there is enough oxygen available for the alkane to burn completely
Incomplete Combustion
Alkane + Oxygen → Carbon Monoxide + Carbon + Water
Note: The incomplete combustion is when there is not enough oxygen available for the alkane to burn completely
Complete Combustion
Alkane + Oxygen → Carbon Dioxide + Water
Note: The complete combustion is when there is enough oxygen available for the alkane to burn completely
Incomplete Combustion
Alkane + Oxygen → Carbon Monoxide + Carbon + Water
Note: The incomplete combustion is when there is not enough oxygen available for the alkane to burn completely
3.5: Describe the substation reaction of methane with bromine to form bromomethane in the presence of UV light
Methane + Bromine + (UV Light) → Bromomethane
Methane + Bromine + (UV Light) → Bromomethane
c) Alkenes
3.7: Draw displayed formulae for alkenes with up to four carbon atoms in a molecule and name the straight chain isomers
Ethene: C2H4
Propene: C3H6
Butene: C4H8
Pentene: C5H10
Ethene: C2H4
Propene: C3H6
Butene: C4H8
Pentene: C5H10
3.8: Describe the addition reaction of alkenes with bromine, including the decolorizing of bromine water as a test for alkenes
Since all alkenes contain a carbon double bond, they will all decolorize bromine. This reaction, therefore, can be used to test for the presence of a carbon double bond (unsaturation) in a molecule.
Since all alkenes contain a carbon double bond, they will all decolorize bromine. This reaction, therefore, can be used to test for the presence of a carbon double bond (unsaturation) in a molecule.
d) Ethanol
3.9: Describe the manufacture of ethanol by passing ethene and steam over a phosphoric acid catalyst at a temperature of about 300°C and a pressure of about 60-70 atmospheres of pressure
Direct Hydration of Ethene
• A mixture of ethene and steam is passed over a phosphoric acid catalyst at a temperature of 300°C and 60-70 atmospheres of pressure:
C2H4 + H2O → C2H5OH
• The ethanol is condensed as a liquid
The ethene required for this reaction is obtained from crude oil
Direct Hydration of Ethene
• A mixture of ethene and steam is passed over a phosphoric acid catalyst at a temperature of 300°C and 60-70 atmospheres of pressure:
C2H4 + H2O → C2H5OH
• The ethanol is condensed as a liquid
The ethene required for this reaction is obtained from crude oil
3.10: Describe the manufacture of ethanol by the fermentation of sugars, for example glucose, at a temperature of about 30°C
Fermentation
• Dissolve sugar/starch in water and add yeast
• Leave the mixture to ferment at room temperature for several days in the absence of air
• Filter off the excess yeast to obtain a dilute solution of ethanol
If the ethanol content is higher than 15%, the yeast is killed. If more concentrated ethanol is required, then fractional distillation is used. The enzymes in the yeast will always produce glucose. The glucose is then converted into ethanol:
C6H12O6 → 2C2H5OH + 2CO2
Fermentation
• Dissolve sugar/starch in water and add yeast
• Leave the mixture to ferment at room temperature for several days in the absence of air
• Filter off the excess yeast to obtain a dilute solution of ethanol
If the ethanol content is higher than 15%, the yeast is killed. If more concentrated ethanol is required, then fractional distillation is used. The enzymes in the yeast will always produce glucose. The glucose is then converted into ethanol:
C6H12O6 → 2C2H5OH + 2CO2
3.11: Evaluate the factors relevant to the choice of method used in the manufacture of ethanol, for example the relative availability of sugarcane and crude oil
The main advantage of direct hydration of ethene is that it is faster
The main disadvantage of direct hydration of ethene is that it requires much more energy
The main advantage of fermentation is that it requires less energy
The main disadvantage of fermentation is that it is much slower
The main advantage of direct hydration of ethene is that it is faster
The main disadvantage of direct hydration of ethene is that it requires much more energy
The main advantage of fermentation is that it requires less energy
The main disadvantage of fermentation is that it is much slower
3.12: Describe the dehydration of ethanol to ethene, using aluminum oxide
Ethanol can be dehydrated by passing ethanol vapor over hot aluminum oxide, which acts as a catalyst for the reaction:
C2H5OH → C2H4 + H2O
This reaction can be used to manufacture ethene for the production of polyethene. Most ethene is made by cracking crude oil.
Ethanol can be dehydrated by passing ethanol vapor over hot aluminum oxide, which acts as a catalyst for the reaction:
C2H5OH → C2H4 + H2O
This reaction can be used to manufacture ethene for the production of polyethene. Most ethene is made by cracking crude oil.